Club Articles
Contents
- Hot Weather Fishing at Shell Island - Eddie Wilkinson, July 2022.
- At Last – The Return to Lake Vyrnwy - Eddie Wilkinson, June 2022.
- Grayling and the River Alyn a short history - Eddie Wilkinson, Jan 2022.
- The Old Rod - Les Jervis, Oct 2020.
- River-fly Population Decline: Suspended Sediment and Orthophosphate - Les Jervis, Oct 2018.
- Conservation Physiology - Recent Catch and Release Research - Les Jervis, Winter 2017.
- Photos of the River Alyn – Spring and Summer 2017 - Mavis Wilkinson, Jul. 2017.
- That little bit of Difference - Eddie Wilkinson, Jul. 2016.
- What do fish from the river Alyn eat? - Les Jervis and Eddie Wilkinson, Oct. 2015.
- Ode to a New Season. - Les Jervis, Apr. 2015.
- Fishing in Cape Verde. - Eddie Wilkinson, Mar. 2015..
- The OTHER Blackflies. - Les Jervis, Mar. 2015.
- Opening Day 3rd March '15. - Eddie Wilkinson, Mar. 2015.
- Blackfly artificials - A challenge for 2015. - Les Jervis, Mar. 2015.
- Simple Sinkant. - Eddie Wilkinson, Feb. 2015.
- Patterns from the Fly Tying Evening. - Eddie Wilkinson, Dec 2014.
- Fly Life on the Alyn - Post Script - Les Jervis, Nov 2014.
- Fly Life on the Alyn - Les Jervis, Oct 2014.
- A Day out with Geoff Clarke - Eddie Wilkinson, June 2014.
- Gammarus pulex – our very own killer shrimp - Les Jervis, Jan 2014.
- Living with Indicators - Eddie Wilkinson, March 2013.
- Mink - Peter Hayes.
- Creeping through the Undergrowth - Peter Hayes.

Hot Weather Fishing at Shell Island
Before lockdown my wife and I went camping on Shell Island near Harlech in Wales. An excellent site mid week outside school holidays !!! The creeks inland at high tide get full of Mullet and occasional Bass they come in with the tide and on our stay in 2018 hundreds if not thousands would follow the tide as it flooded the creeks. I was determined to catch a Mullet on fly, after all how hard can it be !! 1st morning I was up early, right on time, it took me ages but I did catch one (honest), but had forgotten my camera ! No issue I thought I will come back tonight and catch again ? My wife came to, I tried, failed and tried again, my excuse was I was a tad late and the main run of fish had passed. My wife suggested she had a go (she can flyfish, but does not enjoy it) 1st cast she gets a “Good” Mullet and very generously passes me the rod to land it, saying she will take pictures, you guessed it, I lost the fish !!! Her comment – Well I am blowed if I am catching you another. My plan was to return the next day, but bad weather put paid to that, so another trip was planned, then another, then Covid (Shell Island closed), but eventually this July we returned ! The Mullet were still there, but numbers were reduced, may be the time of year ? Also on the 1st day quite a small tide. The best area I found was the causeway, as it is a bottle neck for the fish, which gives me more fish to target and they come in very close. I of course had my “Stealth Gear” on so I could get close without alerting the fish.

Well the 1st day I did struggle. I was using 2 variation of the Romley Shrimp (basically a red tagged shellbacked shrimp) in size 12 (last time I was using an 8). The weather was hot and the water flat calm despite the tide. On the 2nd morning I was up early for the slightly higher tide, absolutely nothing moving on the run, but on full water a few shoals could be seen moving !! I just couldn’t tempt any Mullet, they were feeding lower down in the water, especially the larger ones so I put on my “River Shrimp” a size 10 very pink shrimp. Unfortunately this worked too well, as before any Mullet could get to it “Others” would take advantage !!


Eventually I did manage a few Mullet, unfortunatey I will not be putting in for any “Rod Caught Records” but at least I can now prove I have caught some. If nothing else I did improve my casting accuracy and to watch how close I drop the flies to the fish (not crabs). I used a 9’ 5w fast action Shakespeare rod with a floating line. Will I go back – Yes, maybe even September, weather permitting, once Schools go back, as it is far to busy in the “Holiday” season.

Eddie Wilkinson - July '22

At Last – The Return to Lake Vyrnwy
It has been 3 years since my last visit but June 10th 2022 was the day. The day before, my wife and I were on a Mazda MX5 meeting with Michel Mazda (the dealership we bought the car from). This is a “ride out”, in Thursdays case 67 MX5’s, starting at Ellesmere Port.


Our morning stop was at the Rogan Estate on the banks of the River Dee at the back of Bala. Apparently, Mortimer and Whitehouse are due to fish this beat for their next series. Just over 150m from the manor house. Very neat and tidy, with a nice “hut” with BBQ. Apparently the river here hold a lot of “good” Grayling and the wading looks excellent.



The 2nd stop was Lake Vyrnwy for Lunch. I had carefully booked an overnight stay!


After lunch most of the other drivers went home. We had a walk to the village and on the way out of the hotel met Paul Morgan (Coch-y-Bonddu books at Machynllerth) He had been fishing, but had struggled to catch, using the same method that I was planning ! – not looking good for me. Friday started with a very nice breakfast, before my wife and I parted company, me to the boat and her to the Spa. I chose the the bottom end of the lake that I haven’t fished for at least 20 years ! This was on the recommendation of the staff, who thought it the best option for a poor day ??? The Lake was well down and the staff expected the “Village” to be in view by July. This did give a major problem, in that the trees would not be overhanging the water in many places. The water at the opposite end from the Dam is shallow and does have a weed bed, so insect life will be better in June (I hope). My plan was to “work” this area and the far bank, hoping for a rise or two, but failing that try under the far bank, hoping for a fall of terrestrials. Unfortunately Lake Vyrnwy has some very “Interesting” winds and wind lanes, so much so that at the far end of the lake, the wind can be in “Any” direction at any given time ? i.e. within 10m you can be drifting the near shore, then be blown out into the middle, then reverse back down the lake and carried to the far bank ? Not good for planning a drift ! This problem was taken away from me

By the time I had loaded up (two batteries and an electric motor) and driven the 41/2 miles to the boat station two other boats were already fishing the lower end of the lake. My only option was to fish back towards the dam ! I set up with a dry size 12 “Shiney” fly with a bright green nymph tied NZ style. I was fishing drogueless just using the electric engine to correct the drift.


Hook – Fulling Mill FM12 70 Living Larva size 12
Thread - Ultra 70 in white (whip finish coloured green with pen !)
Tail – Coc-de-Leon 3 or 4 fibres.
Rear half of body – dirty grey Poly yarn.
Front body – CDC split thread wound forward to eye in black.
Hackle – Black genetic cock, wound dense.
Thorax ? – Split the hackle underneath with “Large” Mirage Tinsel.
It took a while but I did get into my 1st Brown within 200m. After a couple more it was apparent that the nymph was not needed. The only problem I had was being in the boat alone as I missed opportunities by drifting out or past fish holding areas. Although no fish were rising, casting the dry within a few feet of the shore brought fish up. If I missed a fish, due to the wind, I struggled to repeat the area. It did not matter the fish kept coming. The “fun” started when I tried to go back ? The little electric motors (37lb thrust) did not make much headway against the wind, but it did get me back to some areas that “required” a second attempt.




By 3pm I had just managed the return to the boat station (all on one battery). I had done well with 16 Browns from a small 6” to an excellent 11/2lb. Most were in the 10oz size but fought well above their size.All the fish were caught on the “Shiney Fly”, including the 6” one. Only used 2 flies, the 1st lasted 10 fish , the second was “ragged” after just 6 ? – Note to self, Tie up some more !!! As room in an MX5 is limited I chose to fish my 7’ 6” ATFM 3 weight river rod with a small collapsible net, all worked well. In previous trips I had gone for my 10’ ATFM 4 weight. I found the change better as I was doing what I normally do on rivers, casting into tight spaces under bushes or close to stones, just felt right. As I write this my plan is to return soon, as I enjoyed the day so much. This happens everytime I go, but I never seem to make it back for the rest of the season, however this year ? Definitely ? Maybe ?
Eddie Wilkinson - June '22

Grayling and the River Alyn a short history
On the “Rossett and Gresford” Club waters
Grayling were not a regular species found until 1999. At this time there were
Grayling below Rossett weir and the Club allowed locals to coarse fish for
them. Some may say that these fish “ran” up the weir or magically flew up over
the weir (in buckets ?). That they began to appear
above the weir in numbers was fact. A little later Grayling began to drift down
from Worms Wood, so we had two sources of these fish. Once in our section these
fish thrived and bred ! Some will remember the little
dimpling rises in the Sewage Pool ? Turned out to be
very small Grayling (less than 11/2”).
Due to the Club being a “Brown Trout River”,
Grayling were not generally recorded in catch returns !
As I kept these “returns” for the club this was my fault. From 2008 the
Grayling had become a major factor in catch returns. They did take a drop in
2013 (for no apparent reason) but recovered strongly. In 2019 there was a 2nd
drop due in most to flooding and lack of fishing opportunities
!

As the numbers returning “catch
returns” have fallen dramatically from 1997 (with a peak of 51 in 2002) to 1 in 2021 it is
difficult to be accurate on “actual” numbers, the one constant has been myself.
Although I do predominantly use nymphs, if I can find rising fish, I will
always use a dry fly and in the past 2 seasons had some great sport on dry’s
including a good number of Grayling.
The table below shows my returns from
2008 when the Club changed the way of recording catches, along with the
“combined” totals for all those who gave in returns. It does not show any major
differences, once members got used to including them !
Trout / Grayling returns 3rd of March to 30th September
Varies on number of visits each season
|
Year |
Brown |
Grayling |
% G to B |
|
2021 |
105 |
104 |
99.04 |
|
2020 |
37 |
77 |
208.10 |
|
2019 |
85 |
56 |
65.88 |
|
2018 |
77 |
101 |
131.16 |
|
2017 |
108 |
142 |
131.48 |
|
2016 |
85 |
116 |
136.47 |
|
2015 |
158 |
119 |
75.31 |
|
2014 |
173 |
77 |
44.50 |
|
2013 |
99 |
34 |
34.34 |
|
2012 |
80 |
85 |
106.25 |
|
2011 |
123 |
87 |
70.73 |
|
2010 |
191 |
65 |
34.03 |
|
2009 |
70 |
80 |
114.28 |
|
2008 |
36 |
81 |
225 |
Combined
Combined totals can be variable, due to numbers submitted and visits, individuals change each year.
|
Year |
Brown |
Grayling |
% G to B |
|
2021 |
105 |
104 |
99.04 |
|
2020 |
85 |
206 |
242.35 |
|
2019 |
232 |
155 |
66.81 |
|
2018 |
134 |
192 |
143.28 |
|
2017 |
222 |
312 |
140.54 |
|
2016 |
151 |
166 |
109.94 |
|
2015 |
117 |
30 |
25.64 |
|
2014 |
530 |
128 |
24.15 |
|
2013 |
402 |
68 |
16.91 |
|
2012 |
403 |
300 |
74.44 |
|
2011 |
356 |
251 |
70.50 |
|
2010 |
416 |
134 |
32.21 |
|
2009 |
380 |
113 |
29.73 |
|
2008 |
282 |
193 |
68.43 |
|
|
|
|
|
For a small river the
Alyn produces some excellent sized Grayling. The best recorded by a member were
3lb 7oz and 3lb 2oz and both came from the same pool. The member had a net with
scales, so both fish were weighed and quickly returned. Many other members have
caught fish in the 1lb to 2lb range, a few have even managed the “magic” 3lb
range.
Most of the larger fish are caught in
the 1st month or so of the season or the last month. This is
dependant on the river levels and when these larger fish decide to move. A few
seasons back these better fish were not seen until the last couple of days of
the season (I had 6 between 1lb and 21/2lb on one section of the river, that
had none in 2 days before !)
Most members do not carry scales and few
if any kill Grayling, so how do you know the weight ?
Below is a chart that may help. For me I measure the fish against my rod and if
possible take a picture. I can then get a measurement
for a “Guestimation” ! Hope it helps.
Grayling
size to weight
Although
this table does not take into acount the time of year
as both male and female will put weight on around girth ready for spawning.
Also note a slight
genetic difference may account for variation.
Gunnar Westrin's table
weight length
0,5 kg 34-38 cm
0,6 kg 38-42 cm
0,7 kg 42-44 cm
0,8 kg 44-46 cm
0,9 kg 46-48 cm
1,0 kg 48-50 cm
1,1 kg 50-51 cm
1,2 kg 51-52 cm
1,3 kg 52-53 cm
1,4 kg 53-54 cm
1,5 kg 54-55 cm
1,6 kg 55-56 cm
1,7 kg 56-57 cm
1,8 kg 57-58 cm
1,9 kg 58-59 cm
2,0 kg 59-60 cm
Eddie Wilkinson.
Jan 2022

The Old Rod
It must have been 1990 or shortly thereafter. I’d moved jobs from Glasgow to Exeter and, with my son Francis, was looking for new waters to fish. In those days, we had only just started to fly fish, most of our time in Scotland having been spent in pursuit of pike and perch. By the time he was just becoming a teenager (some say he was born a teenager, and still is one!) and getting bored with watching floats. He suggested we try fly fishing so a trip to an angling shop in Ayr resulted in two cheap fly rods, second-hand reels, lines and a few flies – mostly Bloody Butchers, the name being a main determinant of choice (I told you he was just starting teen years). We were recommended to try a small reservoir just south of Ayr that was stocked with brownies. A couple of trips eventually brought the desired result but, in retrospect, a bit of tuition would have helped. Our previous experience of casting dead baits and spinners for pike resulted in every self-respecting trout within earshot swimming speedily to Dumfries
I digress but after a couple of years I was offered a job move and Francis wanted a better climate so we all headed down to Exeter with wife and mother in tow. Our fishing became more varied, and successful. One evening, Francis was looking through the local paper and spotted an eight-foot Hardy rod advertised. The phone was immediately brought into service, an appointment made to view and off we went. The old gentleman selling the rod was no longer able to fish and was quite glad to agree a sale. In truth, the rod was in a sorry state. It had been varnished, very badly, and blobs of thick, dark brown, tar-like finish covered the whole length, including part of the handle. The rings were badly worn, the wooden reel seat split and, worst of all, the middle section was fractured. The fracture had been splinted and wrapped but looked terminal. Regardless, a five-pound note changed hands and, with thanks and best wishes, we departed, owners of a Hardy split cane rod of unknown vintage. With job and school pressures, the rod was put away and essentially forgotten. One positive fishing development though, Francis went to a juniors’ fly fishing course in Salisbury, fishing on the Avon. Meanwhile, I had a day on the Wylye at Langford, caught my first grayling and fell in love, shortly afterwards joining the Grayling Society.
Fast forward about twenty years. Retirement for me and a house move to Chester to be close to family. Entirely serendipitously, also moving to live close to one of the best grayling rivers in the UK – the Welsh Dee and one of its tributaries, the river Alyn that flows through Rossett. Meanwhile, the old rod slumbered on. It was not until the Autumn of 2018 that I thought about it again. I had been out-bid for a cane rod at the Grayling Society AGM auction. Driving home I remembered the old rod and dug it out to see if it could be resurrected. As I had first thought, the middle section fracture was indeed terminal. The rest of the rod though looked OK. Worth trying to resurrect? Something to do on long winter evenings, other than tying flies!
Stripping everything down was easy and, under the layers of clotted varnish, the cane looked good. I took off the remains of the handle and reel seat and the brass butt cap. It was a Hardy rod sure enough – “Hardy Bros, Alnwick, Makers” and a serial number – 947 - stamped on the brass butt cap. Must chase that up sometime but now, get new snake rings, cork handle and reel seat fitted. Also to be addressed was the fracture. Getting a new mid-section was feasible but first, I took a hacksaw to it and cut across the fracture. Easy enough to get a set of ferrules to fit. The original twist and lock ferrules were a bit worn but some candle wax tightened them up, at least until I could test the rod on a fish. Putting everything together and finishing with several coats of varnish, the rod looked good enough to cast. I tried a few reels and lines and ended up with an Orvis Battenkill and a 4-weight dry line. Trying it out gingerly on the back lawn, everything held and the rod cast a nice line. A trip to a local stillwater confirmed the casting was OK and the line about the correct weight. Next stop – catch a fish. Not any old fish though, it just had to be a grayling and it had to be on a dry fly.
Unfortunately, a house move intervened. What with packing, unpacking, tidying etc. etc. the winter just flew by and it would be March 2020 before I could hope to get onto the river. However, the time was not completely wasted. I contacted Hardy and got a helpful reply from David Holder. From the serial number – if it was correct – the rod pre-dated 1890. I was staggered. If the rod really was 130 years old, that is almost twice my age. It obviously deserved gentle, respectful treatment. To fish with it or just hang it on a wall? Eventually, out of respect for the old man who sold it to me, I decided I must catch a fish on it. I was sure he wouldn’t want it treated as a museum piece.
Back to March 2020 – and Covid 19 on the horizon. Needless to say, lockdown intervened before I could try the rod. Fast forward again (although it felt like forever) to August. August 22nd to be precise and a trip to the lovely little river Alyn with its good head of grayling. The weather was goodish, overcast, a few rain showers but very breezy. After a spate mid-week, the river was dropping nicely. Arriving at the river I couldn’t see any fly hatching but there were some fish rising at the head of the first pool. On went a parachute black gnat, a fly I’d raised fish with the week before. First cast brought a splashy rise from a tiddler that shook off before landing. A couple of casts later, and a couple of missed takes, a firm take and fish on. The rod handled well but I was very wary about putting too much pressure on it. Eventually a small sea trout came to hand. Not the fish I was aiming for but very welcome and a nice fish to christen the rod with. Rod still OK, no sign of any damage.
I changed the fly to a small light tan Klinkhamer and had an immediate take from what proved to be a small grayling. Target achieved! Both fish were small, about half a pound, but the rod did not delaminate and held the fish well. A heavy shower persuaded me to put the old rod back in the car after a good wipe down. Well pleased, I had a cup of coffee then returned to the river with a nymphing rod. The rest of the session went well but the highlight was those two fish to the old rod, and on dry fly. I was pleased and I hope the old gent who sold it to me all those years ago was looking down and smiling.
Next outing for the rod - the Alyn again - this time I would be more use to the rod. Both casting and striking would be more familiar and I’d have more confidence in putting pressure on fish without worrying too much about imminent delamination. September is usually very good on the Alyn, with lots of fly life and the fish eager to feed in preparation for winter and spawning. Friday 4th September arrived fine, dry but a stronger, much colder, breeze than last time, with the river at a good level after mid-week rain, fining down but with still a lot of colour. A small black Klinkhamer quickly accounted for two grayling around the half pound mark but the increasingly cold and strong wind put down the majority of fish and there were no further rises. I didn’t want to put on a nymph so I changed rods and put thoughts of dry fly fishing to one side.
The Welsh Dee next where some serious grayling should test it more rigorously. The lovely Groves Farm beat seemed ideal. I knew of one spot particularly where, under favourable conditions, the grayling were free-rising. Thursday 10th September was the chosen date. The river had been too high for me to wade safely but by the Thursday, the level had dropped and the water had fined down to a lovely clear peaty colour. It was still higher and faster than I would have liked though for safe wading. Might get something on the dry but nymphs probably a better bet. There seemed to be a few rises close to the bank in shallow water, probably trout or salmon parr but nothing serious was showing. I set up the old rod as for the Alyn with a small black Klinkhamer on the tippet. I had to wade out a bit and the rod would have to cast a longer length than on the Alyn but I soon found fish willing to rise and a grayling of about 12 oz. was soon brought to hand. The rod handled beautifully. It had a lovely curve as the grayling used its dorsal fin well in the strong current. Several more fish of a similar size were soon persuaded to take the fly. When the takes dried up, I changed the fly for a caddis emerger with a curved hook, two CDC feathers for wings and a grizzle hackle for legs. Two more grayling soon obliged.
I decided to have a sandwich, coffee and meet up with Martin who was fishing spiders further upstream. It was the first time Martin had seen the rod so he had a few casts while I ate a piece of his delicious quiche. Martin had no rises but his judgement was that he was surprised how well the rod cast. However, he found the rod heavy and the action slow by comparison with his carbon rods. I think these are part of the charm of cane rods. They make you slow down and make fewer casts but try to make the casts count. When it comes to fish handling, I don’t think there is any real comparison with modern carbon, graphite or glass rods – the rod just feels alive. I guess I’m ‘old school’ as one of the first rods I made from scratch 60 years ago was a split cane spinning rod for pike. I loved the action then and I still do. I still have that rod, which, I suppose, is getting quite old itself. John Gierach writes of the pleasure and charm of cane rods far better than I ever could.
After a catch-up chat with Martin, I decided that I must catch a few more fish before heading home so back to my favourite pool. Whilst wading to a suitable position, I took a tumble and an unscheduled cold bath! This brought my session to an early close. Fortunately, I had a full change of clothes and a towel in the car but it was a very soggy trudge back. The rod, fortunately, survived the ducking.
I’ll improve the rod a bit more by replacing the old twist and lock ferrules over the winter. A bit of candle grease helps but the twist and lock ferrules work loose and don’t maintain a tight enough fit. I’ll keep the rod for dry fly/emerger fishing and try to keep it for fishing on open bits of river. I love the Alyn but there are many trees, bushes, long grass, barbed wire fences and other snags desperately trying to snag a careless back cast and break the rod on the forward cast.
Finally, thanks to the old gentleman in Exeter. I’m just sorry it took so long, but, I got there and the rod is a delight. I still struggle to believe it is 130 years old, but stamped brass rod butts don’t lie, even if they can be transferred between rods. Whatever its true age, the rod is old, at least as old as me (don’t ask!) and probably a good deal older.
Acknowledgement: Thanks to fellow RGFFC member Martin Harrison for reading the draft and for his company fishing over the past few years. Martin, I’ll follow up on your recommendation that I buy a life jacket. I’m getting too old to fish alone without one.
Les Jarvis
Oct. 2020

Originally
published the “Grayling” the journal of the Grayling Society
Volume 27,
No. 8, 26 – 31, Autumn 2018
Reproduced
with permission of the editor
Putting
Research Findings into Practice
River-fly
Population Decline: Suspended Sediment
and Orthophosphate
Les Jervis –
Grayling Research Trust Trustee
Populations
of river-flies have been in dramatic decline in recent years. After many years
of slowly falling numbers, the situation seems to be getting much worse. So
much so that the topic was covered on Radio Four’s “Today” programme and
featured in national newspapers. The Guardian covered the topic under the
headlines “Warning of 'ecological Armageddon' after dramatic
plunge in insect numbers” and “Insect
declines: new alarm over mayfly is ‘tip of iceberg’, warn experts”.
This article summarises some of the
current research being carried out on two of the many factors – suspended
sediment and orthophosphate – involved in the decline of fly life here in the
UK and elsewhere on continental Europe. Germany, in particular, has recorded a
75% decline in total flying-insect biomass in 27 years, even in nature reserves.
There appears to be no escape from whatever is causing the decline. Not only
river-flies but terrestrial flies of all species are suffering the same
dramatic, potentially disastrous, population falls. With the fall in insect
numbers, insectivores – fish, birds, hedgehogs, bats - are increasingly under
threat.
Finding the ultimate cause is proving frustratingly
difficult. Pretty well every possibility has been, or is being, considered from
insecticides through to climate change. Whilst any one factor may be shown to
be involved, multi-factorial systems are extremely difficult, as usually only one
or two factors can be covered in any laboratory experiment. By contrast, flies
in the environment are subjected to vast and complex arrays of probably
constantly-changing and interacting factors. Hence, in any one situation, the
main factors at play probably differ from those in another situation. I will focus
on just two issues here – phosphate and sediment. The papers covered are recent
publications only as the full literature on phosphate and sediment is vast.
In spite of the difficulties, some
progress is being made and just a few of the many recent articles will be used
here to illustrate what is going on and how much more needs to be done.
Comprehensive reviews of the effects
of flow variation, elevated phosphorus and fine sediment have been published
recently by Salmon and Trout Conservation (S&TC), together with a report of
their Riverfly census. What this means for individual rivers is amply
illustrated in the recent press release from S&TC:
Orthophosphate
A very recent study by Everall, Johnson et al looked at the effects of fine
sediment and orthophosphate levels on the early stages of Serratella ignita – the blue-winged olive (BWO) beloved of trout,
grayling and fly fishermen. This paper was covered briefly in the Spring/Summer
2018 edition of “Grayling”. By exposing freshly-laid S ignita eggs to different conditions of suspended solids (SS) and
orthophosphate (OP) individually and in combination, the authors were able to
demonstrate that SS and OP together had a greater effect than either alone. SS
in isolation had a greater effect than OP. Egg mortality in control treatments
was around 6% compared to 45% in treatments with 25 mg/litre SS and 52% in 0.3
mg/litre OP treatments. Even relatively modest levels of each stressor (10
mg/litre SS; 0.1 mg/litre OP), below national legal thresholds, had significant
effects on egg survival to hatching.
Interestingly, when river levels of OP were
measured by the European Environment Agency (EEA), 29 of 72 measuring stations,
(40%) in the UK had levels of OP classed as 5 or 6 (between 0.2 and in excess
of 0.4 mg/litre), well above those used by Everall et al and well above the standards for river orthophosphate levels
set by the Water Framework Directive (Table 1). Very worryingly, this is a higher figure than Germany with 16 of
125 (12%) measuring stations classified 5 or 6 where the 75% decline in fly
populations in 27 years was recorded. Also, very worrying is the fact that
these levels are of annual mean values so highs and lows are hidden. Should high levels
occur when invertebrate eggs are hatching, the mortality rates are likely to be
much higher.
As Everall et al state, “Although levels
are dropping across Europe - substantially in the case of OP - the results of
these experiments support growing concern about current guidelines - - - - and
the need for more stringent regulation”.
Table
1: WFD Orthophosphate Standards for Rivers
|
Existing WFD
standards |
Annual mean of reactive phosphorus
(mg per litre) |
||||||||
|
High |
Good |
Moderate |
Poor |
||||||
|
Existing |
New |
Existing |
New |
Existing |
New |
Existing |
New |
||
|
Lowland, low alkalinity |
.030 |
.019 |
.050 |
.040 |
.150 |
.114 |
.500 |
.842 |
|
|
Upland, low alkalinity |
.020 |
.013 |
.040 |
.028 |
.150 |
.087 |
.500 |
.752 |
|
|
Lowland, high alkalinity |
.050 |
.036 |
.120 |
.069 |
.250 |
.173 |
1.000 |
1.003 |
|
|
Upland, high alkalinity |
.050 |
.024 |
.120 |
.048 |
.250 |
.132 |
1.000 |
.898 |
|
|
Everall et al experiment levels |
0 – 0.3mg per litre |
||||||||
|
EEA levels |
Class 5: ≥ 0.20
< 0.40 mg/litre |
Class 6: ≥ 0.40
mg/litre |
|||||||
In a recent study, Withers et al examined the effects of reducing
agronomical optimum soil phosphorus levels. They concluded that these optimum
levels still posed a eutrophication risk and that innovations in
agro-engineering of soils, crops and fertilisers could reduce soil phosphorus
levels and with it the risk of eutrophication, without affecting productivity adversely.
They recommend making soil phosphorus testing compulsory in priority catchments
suffering, or at risk from, eutrophication ensuring that good fertilizer and
manure management fully account for soil phosphorus. Rowe et al point
to the value of legacy phosphorus, accumulated in soils from previous
applications. If properly managed, this could potentially replace much of the
phosphorus added annually from diminishing finite resources of phosphate,
without affecting soil productivity, reducing eutrophication and making
nutrient management more sustainable. Taking this further, Withers et al focus
on minimizing phosphorus input by recovering and recycling phosphorus and
avoiding excess run-off into water courses.
Suspended
Fine Sediments
This is a major cause for concern,
suspended material can be either inorganic – clays, soil etc. or organic –
sewage effluent, farm slurry waste, and either can bind soluble materials like
phosphate and complicate interactions with the aqueous environment.
A recent special issue of the journal
“River Research and Applications” was devoted to the effects of fine sediment
on rivers. The introductory chapter by Mathers concludes that “understanding
and mitigating the effects of fine sediment pressures remains an important and
multifaceted problem that requires interdisciplinary collaborative research to deliver
novel and robust management tools and sustainable solutions”.
The issue is a collection of papers on
methods, case studies and implications. Of the papers, those by Béjar et al, Sear et al, Collins et al and Extence et al probably
illustrate the scope of material covered in the special issue.
The paper by Béjar
et al
took advantage of a natural situation to isolate the effects of suspended
sediment (SS) whilst other factors such as flow rate are kept constant. By
following the effects of short periods of river gravel extraction in the River Cinca
in the Pyrenees, they were able to follow changes in the drift of different
invertebrate species before and after the SS changes caused by the resulting
sediment load. Of the species looked at, Ephemera,
Plecoptera and Trichoptera all
showed increased drift whilst Chironomidae
showed decreased drift. The authors suggest that this may be due to the
occupation of different zones by the different invertebrates. They point out
that the River Cinca experiences repeated cycles of periodic high sediment
loads, with subsequent storage of sediment on the river bed and then subsequent
river bed mobilisation and that rivers only rarely experiencing this sequence
of events may respond differently. However, the Cinca situation is likely to be
common. The authors also suggest that different invertebrates may also differ
in their drift response. Ephemera danica,
with a burrowing nymph, may differ from Baetis
spp, which have an agile darter nymph living on the surface of the bed and
more prone to flow and other disturbance. The majority of natural increases in
SS will coincide with increases in river flow after spates and floods, which
will increase shear forces and bed mobilization, both likely to increase
invertebrate drift, whereas the Béjar et al paper
isolates SS increase from these complicating factors. River flows will also
affect the concentration of other factors such as orthophosphate, pesticides
etc., reducing their concentration as flow increases. At the same time, floods
and spates may wash off additional pollutants depending on recent activities on
bordering fields, thereby introducing more stressors along with suspended
sediment. These are yet
further complicating issues when it comes to designing experiments that mimic
real situations. These complex issues point to the need for, and adoption of, new
biomonitoring techniques and new experimental techniques allowing investigation
of simultaneous multiple stressors.
In
addition to SS effects, Sear et al
illustrate the importance of sediment-associated organic material (OM) in
respect of its effects on hatching salmon eggs. The additional oxygen demand
from OM, combined with the decreased oxygen diffusion due to reduced
inter-gravel flow due to sediment infiltration of interstitial spaces, caused
delays in hatching but little effect on overall egg mortality. Such OM substantially
increases the sediment oxygen demand (SOD). Although Sear et al did not identify the origins of the OM in their experiments,
a follow-up paper by Collins et al
did so in a paper that looked at fingerprinting source material in the River
Axe, and found that farmyard manures, septic tank and waste water plant
effluents, decaying vegetable matter and damaged road verges all contributed in
varying amounts. The authors point to
the effects that these can have on
critical
periods of juvenile fish development and suggest that reconnaissance surveys
for OM
sources and SOD should be extended with sample collection linked to these periods. They do not specifically mention
invertebrates but the same factors will apply as will the suggestion for
improved reconnaissance.
Extence
et al, reported a series of case
studies using the “Proportion of Sediment‐sensitive Invertebrates (PSI) Biomonitoring” index. The PSI
score describes the proportion of fine‐sediment‐sensitive
invertebrates in samples taken by timed
net/sweep surveys, with different habitats being sampled with effort
proportional to their occurrence. Scores range from 0 (entirely silted river
bed) to 100 (entirely silt‐free river
bed). The authors have applied the PSI scoring system across the entire of
England and Wales using EA and NRW data. Using a small number of case studies,
they were able to demonstrate that, in rivers like the Ancholme that
is not achieving good WFD status for phosphate, diatoms, macrophytes, and fish,
all of these failing elements can be linked to sedimentation. Nevertheless,
separating the effects of sediment from those of flow is difficult.
Recent work on macroinvertebrate biological traits has
looked at whether these traits can be used as indicators of stress and indicate
the degree to which stream gravel bed interstitial spaces are filled with fine
sediment. The research group made 18 predictions for how macroinvertebrate
traits would respond to fine sediment stress. Only 2 of these predictions were
supported by the results of findings. One was that invertebrates that give
birth to live young, such as Gammarus
pulex, would increase in prevalence whereas swimming and crawling
invertebrates, such as Baetis spp.
would decrease. The proportion of scraping invertebrates (such as Ecdyonurus spp) also declined with
increasing sediment as stones become covered in a layer of silt.
Multiple Stressors
and Mesocosms
Many of the more recent reports use
mesocosms to more accurately represent genuine river conditions. It is difficult to replicate field
conditions in the laboratory and to properly identify the individual effects of
different factors, artificial single or double component systems are preferred.
Artificial mesocosms allow the study of multiple stressors and their
interactions and these are being
using more widely in recent work. A short video showing the experimental set-up
can be found at: https://www.youtube.com/watch?v=a67G0GH9COs. Using such a system, Piggott et
al
manipulated nutrients (simulating agricultural run‐off) and deposited fine sediment (simulating
agricultural erosion) and water temperature simultaneously in 128 streamside
mesocosms. They found that all three stressors had pervasive individual
effects, but in combination often produced additive or antagonistic outcomes. Diversity and structure of Ephemeroptera, Plecoptera
and Trichoptera (EPT) (Insecta) assemblages, used worldwide to measure stream
health, showed complex three-way interactions. High temperatures exacerbated
sediment effects suggesting climate warming will lead to further degradation of
already impacted streams and rivers. Invertebrate drift was increased by
suspended sediment for many species but chironomid larvae and burrowing nymphs,
such as those of E. danica, were not
affected. This is in good agreement with the results of Béjar et al.
Jackson et
al analysed 88 different research papers and concluded that the effects of
multiple stressors could be antagonistic, synergistic, additive or reversed.
They concluded that there was a need to lessen the exposure of aquatic systems
to stressors.
Improvements
to Agriculture and Land Use
Whilst there is some good news – the new rules for farmers and land
managers to prevent water pollution (https://www.gov.uk/guidance/rules-for-farmers-and-land-managers-to-prevent-water-pollution) - much remains to be done
and the effectiveness of new measures will need close monitoring, both by the
scientific community and by anglers.
In a recent publication, Doody et al stated that “Despite widespread implementation of best management practices,
sustainable farming is neither practical nor possible in certain locations,
where protecting water quality and promoting agricultural production are likely
to be incompatible. Some strategic prioritization of land-use options and
acceptance of continually degraded waterbodies may be required to ensure
optimization of multiple ecosystem services in catchments (also known as
watersheds or drainage basins).” In spite of sounding rather ominous, the
paper attempts to set out “a flexible
conceptual framework that can account for catchment sensitivity to multiple
anthropogenic pressures” With ever-increasing demands on agriculture and
the resulting intensification of farming, a rational approach to the management
of land use and water quality is sorely needed. The international group of
authors, which includes Professor Paul Withers of Bangor University, propose a
catchment buffering capacity approach, whereby the ability of agricultural land
in a catchment area to act as a buffer between input of fertilisers etc. and
leaching into rivers is determined to assess the capacity to sustainably
intensify agriculture in the catchment. The authors give some interesting
examples. In New Zealand, deer farming in thin soil upland environments near
river headwaters has a higher phosphate export than lowland dairy farming in
catchments with deeper, better-buffering soils. Two catchments with similar
agricultural intensity can have very different outcomes in terms of impacts on
aquatic ecosystems. They also point to the example of Lough Melvin in Northern
Ireland where, in spite of low-intensity agriculture and mitigation efforts,
there has been a substantial decline in water quality. These, and other examples,
point to the need for new methods for determining catchment buffering
capacities and the threshold points beyond which catchment buffering fails.
The authors discuss the buffering capacity
concept in the context of agricultural use of phosphate but state that it
should also be assessed for its applicability to other stressor such as
sediments.
In a paper that looks at agri-environmental
schemes, Jones et al, analysed data from Welsh schemes designed to improve the
interactions between agricultural practice and environmental improvement. The
authors point out that the European Union spent 23 billion Euros between 2007
and 2013 on agri-environment schemes (AES). Yet there has been little real
evidence that this has resulted in any improvement in water quality because, as
the study points out, results were monitored on an input approach – what
actions farmers took – as opposed to an outcomes approach – what was actually
achieved.
The authors analysed the Welsh AES data based on water quality and
freshwater ecosystem condition and concluded that it was very difficult to
demonstrate positive outcomes by simply comparing scheme areas with non-scheme
areas. Although the analysis was hampered by a lack of long-term chemical
analysis data, by using the outcomes-based approach and modelling diffuse
pollution and biological indices, they were able to demonstrate strong links
between AES and reduced diffuse pollution.
They suggested a targeted approach, spending money to reduce pollution
where it is most needed. The subsequent Welsh Glastir AES was the first in
Europe to adopt this targeted approach and to measure the success of the scheme
through outputs, and the approach is now used in England and elsewhere.
Although intensification of farming
contributes very significantly to both orthophosphate and sediment problems, it
is far from the only contributor. Anyone watching the BBC Countryfile programme
on 24th June, 2018 will have seen the extent to which water
companies use CSOs – Combined Sewage Overflow discharges. In spite of being
warned by central government to have full monitoring of CSOs in place by 2020, many,
very profitable, companies are way behind schedule. The use of so-called
emergency discharges via CSOs is becoming an unmonitored routine and even the
dischargers don’t know, or claim not to know, how much or how often they are
used. Without monitoring being fully in place, anyone seeking to challenge the
over-use of CSOs is at an immediate disadvantage. It is unlikely that water
companies will take pre-emptive corrective action. Without successful legal
action and punitive penalties, they will continue their current course of
inaction. The extent of pollution caused by single septic tank overflows can be
considerable. Regular use of CSOs can be devastating and confounds water
quality monitoring and attempts to reduce diffuse pollution by improvements in
agricultural practice.
Attempts are being made, supported by funding
from water companies, to develop hydro-ecological models that predict the
long-term negative effects of water abstraction on macro-invertebrates. These
attempts show the difficulty and complexity of such vital work but have shown
that macroinvertebrates were not resilient to long‐term, steady‐state
levels of groundwater abstraction when flows at Q75 (flows that are achieved
75% of the time under normal conditions) were reduced by more than 50%.
Buffering
Capacity Thresholds and Environmental Tipping Points
An
environmental tipping point has been defined as “a
part of the human-environment system that can lever far-reaching change in the system. A change at the
tipping point sets in motion mutually reinforcing feedback loops that propel
the system on a completely new course”.
The link between tipping points and buffering capacity thresholds seems
evident: beyond a catchment’s ability to buffer input, the resulting increased
output into rivers and lakes will sooner or later push the ecosystems in those
water bodies over the tipping point, changing and reducing ecosystems as the
process of eutrophication increases. The catchment buffering capacity system
would prioritize either agriculture or aquatic ecosystems. As Doody et al state “Alternative water-quality objectives can be set
if the cost of achieving a desired target is deemed unduly expensive or if a
human activity supported by a waterbody is considered essential
to society”. Further
work is required “Disentangling the
effects of these multiple stressors at the catchment scale remains difficult, but
is a vital step toward prioritizing catchments”. Whatever the future holds,
it seems inevitable that difficult decisions will have to be made and the
degradation of some water bodies probably accepted in the trade-off between
intensive agriculture and water quality.
Rules for
Farmers
Imposing “Rules for farmers and land managers to
prevent water pollution” by DEFRA are likely to have little effect as “farmers require proper educational and extension
services to respond to the challenges of sustainable intensification, with
adaption to precision nutrient management unlikely if farmers do not have
adequate access to extension services with trained personnel” (Doody et
al). The Environment Agency is very rarely given sufficient resources to deal
with all the requirements placed on it. It seems unlikely, therefore, that
sufficient trained personnel will be available to deal with farmers’ requests
for help in a very complex situation. However, advice is available through the
Catchment Sensitive Farming Initiative. In addition to rules for farmers, UK
Government should issue rules for water companies. They are profitable,
well-staffed and do not need the level of support that farmers require. They have
no excuse for inaction, although their inaction over leaking infrastructure has
been evident for years with hose-pipe bans introduced as a first-choice
alternative to remedial action.
Although there are improvements in monitoring
methodologies and remediation techniques, methods for the re-introduction of
invertebrates may be necessary. The Game and Wildlife Conservation Trust (GWCT)
has issued its GWCT Guide to Farming Habitat Issues that covers pretty well
every aspect of farming for improving habitat for wildlife, much of which
requires insects for food. The GWCT also sent a letter to the Guardian about
its own long-term studies into insects on farmland which found a 72% decline in
insects between 1970 and 2015, with strong indications that this was due to
insecticide use. Recommendations for farmers included establishing
“conservation headlands”. Other guidance is issued by the NFU aimed at, for
example, encouraging and protecting pollinating insects.
In addition to improvements in monitoring
methodologies and remediation techniques, methods for the re-introduction of
invertebrates are necessary. The Game and Wildlife Conservation Trust (GWCT)
has issued its GWCT Guide to Farming Habitat Issues that covers pretty well
every aspect of farming for improving habitat for wildlife, much of which
requires insects for food. The GWCT also sent a letter to the Guardian about
its own long-term studies into insects on farmland which found a 72% decline in
insects between 1970 and 2015, with strong indications that this was due to
insecticide use. Recommendations for farmers included “conservation headlands”.
Other guidance is issued by the NFU aimed at, for example, encouraging and protecting
pollinating insects.
The majority, if not all, the issues raised
cannot be dealt with by individuals but, as members of local angling clubs etc.,
we can all try to influence local riparian owners, farmers, the EA, NRW, SEPA,
water authorities and any other relevant body. All possible sources of
pollution need to be monitored and, where necessary, problems addressed. If we
don’t do the necessary nagging, no-one else is going to. At the national level,
GS, GRT, WTT, STC etc. need to keep up pressure on government departments,
ministers and agencies, particularly as Britain leaves the European Union and
WFD.
The links between river ecosystems, including
fly-life, are very difficult to disentangle.
Attempts have been made to agree lists of
priorities in respect of enhancing biodiversity monitoring and the introduction
and standardization of new and improved methodologies are high on the list that
was agreed by 52 experts.
Most,
if not all, of our rivers are suffering from a shortage of fly life and, with
it, a dearth of rising fish. The majority of the population do not notice,
except that drivers no longer have to clear car windscreens of dead insects
during journeys, bird watchers observe fewer and fewer insectivorous birds, and
bat and hedgehog numbers continue to decline. If the decline of both aquatic
and terrestrial flies continues, there will soon be only vanishingly small
numbers left.
It is
easy to blame the intensification of farming or water abstraction or sewage
pollution for the declines but doing so offers no immediate solutions. Given
the choice, any Government will choose secure, adequate, food and water
supplies and waste disposal for voters against the environmental health of
rivers and land – however short-sighted these choices might be. After all, Governments
only last for 4 years and long-term thinking is not their priority. Without
another “Great Stink of 1858”
shutting down Government, it is difficult to know how the attention of
Parliament can be focused on a shortage of fly life. Each of us must try,
however futile it may seem, to influence those who can act. With Britain
heading rapidly towards Brexit, we shall soon fall out of any protection,
however tenuous, that the Water Framework Directive and other EU environmental
protection measures offer. New legislation has been promised and, hopefully, it
will continue with the policy direction defined by the WFD. it is to be hoped
that relevant MPs are effective in putting in place strong measures to replace
them with something at least as effective. Without some urgent, Europe-wide,
action it is very difficult to conclude that we are not at an environmental
tipping point that extends far beyond the health of rivers, their fly life and
rising fish.
The
problem cannot be ignored or left to the attention of a few enthusiasts such as
Dr. Cyril Bennet. His recent work on the Test should be read in conjunction
with the final quote (below) to see the truly shocking degradation that has
occurred in a single century. It is immediately obvious who the most disastrous
enemy of fly life is.
Politics
is supposed to be, in the words of Otto von Bismark, “the art of the possible”.
It ought to be possible to prevent this remorseless decline.
If the Editor will allow me one last
quote, I can add nothing further: “Thicker
and thicker grew the mass of fly over the water, more and more numerous those
carried down by the current. In the backwaters and eddies they were packed
nearly solid. In the main current, they were separated only by inches. All the
broad river was covered. Now all these that had escaped the attacks of trout
and grayling, and swifts and swallows and martins and wagtails and warblers and
chaffinches and many other birds which prey on them, all of them had escaped,
and reproduced their species; when you looked at the countless thousands which
floated down in the small time during which you saw only a small part of the
river, you realized that the quantities of them which had survived were so vast
that the assault of all their enemies made no appreciable impression on their
numbers”.
From John Waller Hills, (1867 – 1938),
“A summer on the Test”, ISBN B002SJIF2Q.
Acknowledgements
Many thanks to Dr. Iwan Jones of the School
of Biological and Chemical Sciences, Queen Mary University of London, Professor Maria Bejar of
the Fluvial Dynamics Research Group (RIUS),
University of Lleida, Lleida, Catalonia Spain, Professor Matt Johnson of the School of Geography,
University of Nottingham, Professor Paul Withers of the School Environment,
Natural Resources and Geography, Bangor University and Professor Adrian Collins of Sustainable Agriculture Science, Rothamsted Research,
Okehampton, Devon, for help with obtaining recent papers and reading the
draft of this paper. Thanks also to other authors mentioned here for copies of
papers and offers of help, the academic community has been overwhelmingly
helpful. My thanks also to Malcolm Greenhalgh, Alan Swann and Rich Cove of
GS/GRT for commenting on the draft paper, and to Mark Pierce of the Welsh Dee
Trust and Rossett & Gresford Flyfishers Club for invaluable help.
Links:
2. https://www.youtube.com/watch?v=a67G0GH9COs.
3.
https://www.gov.uk/guidance/rules-for-farmers-and-land-managers-to-prevent-water-pollution
4.
https://voluntaryinitiative.org.uk/water/catchment-sensitive-farming/
5. https://www.gwct.org.uk/media/483353/farminghabitatguide.pdf
7.
https://www.gwct.org.uk/farming/advice/habitat-issues/conservation-headlands-field-margins/

Originally
published the “Grayling” the journal of the Grayling Society
Volume 27,
No. 6, 10 – 13, Winter 2017
Reproduced
with permission of the editor
Putting
Research Findings into Everyday Angling Practice
Conservation
Physiology - Recent Catch and Release Research
Les Jervis –
Grayling Research Trust Trustee
The Grayling Research Trust is
supported generously by Grayling Society members via annual subscriptions and
the auction that takes place at the AGM. In addition to funding research into
various aspects of grayling, including genetics, ecology, habitat, fisheries
management etc., the GRT supports
students studying for MSc and PhD degrees. These are important, not only for
the insights they bring to our understanding of grayling biology but also for
building future research capacity.
Some of the research carried out on
grayling leads to changes in the management practice of grayling fisheries in
the UK. For example, the GRT co-sponsored research into UK grayling population
genetics has eventually led to changes in grayling stocking policy advice given
by the Environment Agency.
Ongoing GRT-supported research into
the changes in grayling populations in the River Wylye over 20 years is leading
to a better understanding of the environmental factors that affect population
changes. Notably these include the effects of spring floods on egg and fry
survival and the consequential changes in recruitment into older specimens and
future breeding. This latter work holds out the promise of a better
understanding of the implications of warming and over-abstraction of water from
our rivers. Indeed, priorities for GRT support discussed at the recent GRT
meeting in Wrexham included setting up another long-term study on rain-fed
rivers, the effects of climate change and of avian predation on grayling
populations.
In addition to the mainly UK research
that GRT supports there is, of course, much more grayling research conducted in
Europe, Asia, Canada and North America. Like the UK research, much of this has
longer term potential benefits. Some however, has implications for immediate
improvements in angling techniques.
One of the most active groups researching
the effects of angling on grayling and other salmonids is that of Professor
Steven Cooke based in Canada but with international links. One such link with
Norway has resulted recently in the publication of the first work on catch and
release practice directed specifically at European grayling. This work is the
focus of this article and details of the paper are given at the end.
Whilst C&R might seem to have
obvious benefits, research has shown it can lead to high mortality and
morbidity. In the case of salmonids, the average post-release mortality has
been estimated at about 15% - 1 in 6 of the fish we catch die after release.
This rate is very angler-dependent, with the best practice having under 1%
mortality and the worst in excess of 50%. These figures obviously impact on
conservation and C&R research is now referred to as conservation
physiology.
With such post-release mortality, it
obviously pays to take note of research findings and put them into our
individual practice as soon as we can. Estimating post-release morbidity is
much more difficult but we have probably all encountered fish with fungal
infections and damaged mouths caused by poor handling and from poor release of
barbed hooks.
Professor Cooke and two Canadian
colleagues, Robert Lennox and
Kim Whoriskey, worked with Norwegian researchers
Ian Mayer, Torgeir Havn,
Martin Johansen, Eva Thorstad and Ingebrigt Uglem from Trondheim and Oslo to
examine the factors affecting the survival of grayling after capture on rod and
line (Lennox et al, 2016). The work
was undertaken on the sub-arctic River Lakselva in August 2014 using 3 – 4 weight rods,
floating line, barbless J hooks, artificial flies and rubberised knotless
landing nets. The water temperature was 17 – 18 degrees. At higher
temperatures, fish would be more stressed and take longer to recover given that
oxygen is less soluble in warmer water. Fifty-two similar sized fish (32 +/-
4cm) were either landed quickly (less than 2 minutes) and used as a baseline
group or fished to exhaustion then
divided into three groups that were subjected to different periods of air
exposure.
The baseline group of fish was held in landing nets for
unhooking and reflex assessment. The other group was exposed to air for periods
of time from 10 seconds to 2 minutes before being returned to the river in
cylindrical bags with mesh at each end, to allow for adequate oxygenation, to
recover. To measure stress responses, fish were kept for up to 30 minutes after
air exposure before blood samples were taken for the measurement of pH, lactate,
glucose and sodium. The results were compared with samples from baseline fish taken
immediately after capture. All fish were assessed for reflex impairment after
capture and after air exposure, an important parameter as reflex speed is
important for predator avoidance.
The main physiological increase in captured fish was
lactate (the compound that makes muscle ache after exercise and, in extreme
cases, seize up) but there was little effect on glucose, pH or sodium. Stress
impairment was about 25% immediately after capture but this rose to about 30%
after 10 second air exposure and 100% after two-minute exposure. Not only was
reflex impairment increased on air exposure, recovery was also prolonged at
exposures beyond 10 seconds and recovery was still incomplete after 30 minutes
resting when air exposure was 2 minutes.
The capture event, when done to less than full
exhaustion, was minimally stressful for the fish. Prolonging the landing of fish
until they were exhausted led to further increases in lactate and recovery
time. Optimum C&R practice would, therefore, be for fairly rapid landing,
unhooking in water without a net or in a non-abrasive rubberised net, and photography,
if necessary, in water or with a very short air exposure.
Perhaps C&R should be modified to C&(Quick)R –
except where fish need to be revived before release because of excessive
lactate acid build-up and/or higher water temperatures.
In other recent papers from the Cooke group, different
aspects of C&R have been investigated. Assisted ventilation to help
captured fish recover before release (Brownscombe et al 2017) showed some, but not statistically significant, benefit
for brook trout from maneuvering the fish in a backward and forward manner to
help oxygenation.
The fish were often found to swim a short distance from
the release point before settling on the river bottom for 10 minutes or more to
recover further. During this time, fish were poorly prepared to avoid
opportunistic predation (mostly by pike). One possibility considered was that
holding the fish during assisted recovery actually increased the stress level.
If fish were actively respiring (opercula opening and closing) any further
assistance did not speed up oxygenation. Unfortunately, the period of air
exposure was long (8 minutes) and recovery was performed in tanks, not running
water. Few anglers would dream of keeping a fish out of water for such a
prolonged period and holding a recovering graying in running water is always the
available option. It was noted that fish recover much more quickly if the mouth
is open, possibly (though not suggested by Brownscombe et al) because flow of water through the buccal cavity helps to
stimulate and regularise the heart rate. This increases blood flow through the
gills, improving re-oxygenation.
Holding the mouth of a fish open whilst keeping it
upright with minimal handling provided the fish is facing into the water
current seems optimal for recovery. The small underslung mouth of grayling
might make this difficult with smaller specimens but should be feasible with
larger, older, fish. This seems an area for further research.
In a forthcoming paper (Lizee et al, in press), the Cooke group look at the influence of landing net mesh type on
handling time and tissue damage of angled brook trout. From already published
work, it seems likely that the damage to fish, particularly lively fish, a
landing net can inflict can be substantial, leading to post-release infection.
My guess is that rubberized knotless nets will be shown to cause the minimum
damage but, where possible, we should probably avoid net use unless necessary
to aid fish unhooking or speed-up landing. It is already illegal in England,
Scotland and Wales to use anything other than knotless nets.
A recent editorial in the
journal “Fisheries Research” (Cooke et al,
2017), summarises the outcomes of a meeting of the American Fisheries Society
(August 2015) to discuss the current state of catch and release science and
practice in the 21st century. One of the more notable patterns to
emerge was that the behavior of individual anglers has a major influence on the
outcomes of C&R. That behavior includes gear selection (e.g. hook type,
bait, leader and tippet strength), time and location of fishing (predator
presence, season, depth) and how anglers interact with the fish post-capture.
The meeting concluded that it is necessary to put the emphasis on the angler
when conducting and applying C&R science – “Inserting the Angler into
Catch-and-Release Angling Science and Practice”.
The Cooke group has also
published a paper recently, (Brownscombe et al, 2017b) summarizing “Best
practices for catch-and-release recreational fisheries – angling tools and
tactics”) from hooking through to recovery techniques. The paper is too long to
summarise adequately here but is freely available on the Cooke Lab website and written in very accessible terms. It merits
reading by all GS members. Many of these practices are summarized on the
“keepemwet.org” website that can be accessed through the Grayling Society
website and which already informs members of the Grayling Society. It will come
as little surprise that Steven Cooke and his colleague Andy
Danylchuk are deeply involved with
keepemwet.org. The keepemwet.org website puts advice in a much better way than
I can but, whatever we do, we have to accept that some fish, however carefully
handled, will die post-release. The best we can do is always treat our quarry
with respect and handle all fish minimally and carefully. As Alan Roe said in
his talk at the recent GS symposium in Wrexham, grayling always give 110% when
fighting. We should give back 110% trying to ensure their survival after
release.
All this applies
to what is best called “leisure angling”. One major problem occurs in
competitive grayling fishing when, in attempts to catch more fish, some anglers
throw-back fish as quickly as possible. Grayling, especially large ones, that
have been stressed by being played out for some time will, when released, turn
belly-up and, unless grabbed gently and held the right way up until fully
recovered, inevitably die when otherwise they would probably survive.
All the C&R research in
the world will fail to influence fish mortality rates and conservation unless
the individual angler puts findings into practice. The difficulty is spreading
best practice outside the constituency of conservation groups like the Grayling
Society. Although the GS has its own Code of Practice, anything we as
individuals can do to improve our own practice and influence fellow anglers can
only be for the benefit of grayling conservation.
Acknowledgments
The suggestion for this
article came out of discussion at the annual GRT meeting held this year in
Wrexham along with the GS Symposium and AGM. Thanks are due to Malcolm
Greenhalgh, Rich Cove, Ross Gardiner and Alan Swann for reading and commenting
on the manuscript.
References
Full pdfs of all the above published papers
can be downloaded free of charge from:
Cooke
Lab Publication Page: http://www.fecpl.ca/publication/
1. Lennox R.J., Mayer I., Havn T.B., Johansen M.R.,Whoriskey K., Cooke S.J., Thorstad E.B. and Uglem I.
(2016).
Boreal Environment Research 21: 461–470, 2016
Effects of recreational angling and air exposure on the physiological
status and reflex impairment of European grayling (Thymallus thymallus)
2.
Brownscombe
J.W., Parmar T.P., Almeida J., Giesbrecht E., Batson J., Chen X., Wesch S., Ward
T.D., O’Connor C.M., Cooke S.J.
(2017a). Fisheries Research, (2017), 186(3), 619 – 624 The efficacy of assisted ventilation
techniques for facilitating the recovery of fish that are exhausted from
simulated angling stress
3.
Brownscombe
J.W., Danylchuk A.J., Chapman J.M., Gutowsky, L.F.G., Cooke S.J. (2017b).
Fisheries Research, (2017), 186(3),
693 - 705: http://dx.doi.org/10.1016/j.fishres.2016.04.018
Best practices for catch-and-release
recreational fisheries – angling tools and tactics
4.
Cooke S.J., Palensky
L.Y., Danylchuk
A.J. (2016).
Fisheries Research Editorial / Fisheries
Research 186 (2017) 599–600
http://dx.doi.org/10.1016/j.fishres.2016.10.015
Inserting the angler into
catch-and-release angling science and practice
5.
Lizee
T.W., Lennox R.J., Ward T.D., Brownscombe J.W., Chapman J.M., Danylchuk A.J.,
Nowell L.B., Cooke S.J.
North American Journal of Fisheries Management:
In Press.
Influence of landing net mesh type on handling
time and tissue damage of angled brook trout.

Photos of the River Alyn – Spring and Summer 2017
In this article we shall start at the downstream limit of our fishing, the railway bridge at Rossett, and move up to our upstream limit at Pont y Capel. The pictures on the left were taken in spring while those on the right were taken in summer.
Railway Bridge to Two Bridges (Start of fishing at railway bridge)
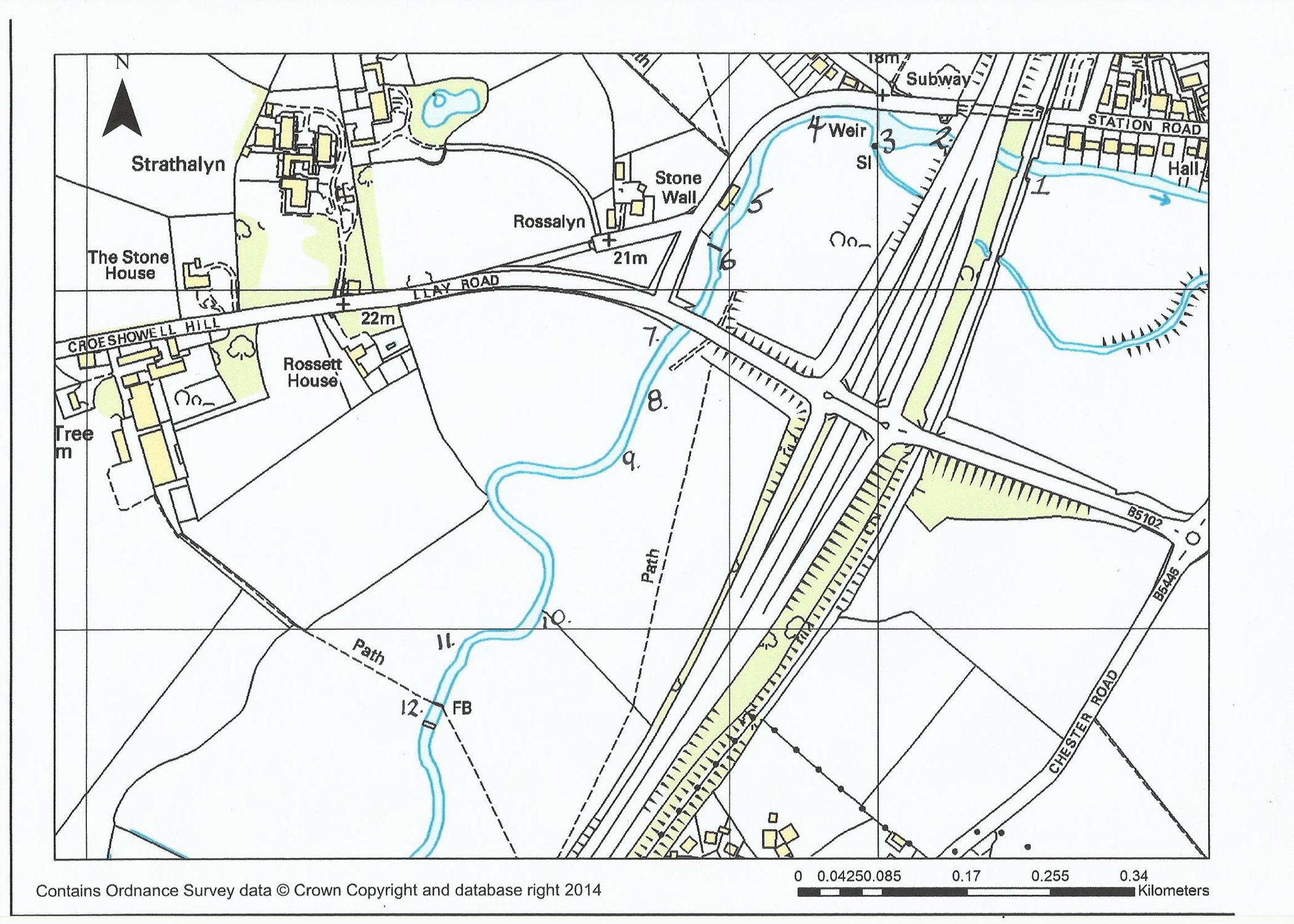
1. Railway bridge to dual carriageway bridge – shallow fast water with odd fish in depressions or by railway bridge columns.


2. Upstream corner of the dual carriageway bridge - deep hole into fast water – good grayling here.


3. 30 metres below and above the weir there is no fishing due to bye-law restrictions


4. Horse Chestnut tree to bend before Riverside Cottage – good grayling with occasional free rising brown trout, bit slow water. Can be fished from the one bank or by wading up the middle (shelf). Too deep to wade this bend, so need to retrace steps to get out.


5. Front of Riverside Cottage to footbridge - difficult area to fish due to access and back casting limitations – good brown trout here.


6. Footbridge to Llay Road Bridge. Good rising browns normally under the weeping willow and up to the island – Right bank.



7. Llay Road Bridge to the island – a mix of rising fish just upstream of bridge to nymph feeders at the island. Mixture of water too from fast shallows to deep holes. At the downstream end of the island there is a channel off the left bank that holds browns, but casting is hard and short but well worth the effort.


8. The pool above the island under the cracked willows is deep water with the odd rising fish – well worth a try. To access the next area cross the river at this point. Above the deep pool there are usually rising fish in the shallows on the right hand side. These tend to be easily put down – deeper water is on the left. Do not ignore the area on the right under the trees. There are some stones across the river – fish up to these.



9. If you are wading get out on the left bank. This takes you to the next pool. Alternatively there is a stile and access route from the field. This pool has regular rising fish normally at the mid water and over to the left bank, in front of the tethered log. Wading the right side is possible but it will kick up silt very quickly and colour the pool downstream. There are several small pools now and some riffles before the bend in the river. This section is not always fished as people ignore the stile and walk along the edge of the field. The bend has a deep water area that shallows off as you go upstream but usually a good place to catch a brownie or two. This area is normally referred to as the cabbage patch because of the large leaf plants in the area. As you move away from the bend you come to a nice pool that has an obstruction and fast water leading into it. This will normally hold quite a few brown trout, but is difficult to fish due to drag.


10. Moving above this area and into the next field there is a straight section that has fronds of felt embedded in the river (a trial by the EA after the floods of 2000). This section leads to a very nice deep pool by the next island, which can be very productive for brown trout.


11. Above this area is another island with a stone revetment on the left bank. The first 20 metres has deep water on the left bank – this moves into riffles and shallows before coming into a really good grayling hole under the power cables, but before the twin bridges.


12. The second of the twin bridges looking upstream.


Although it looks good between the two bridges it can be difficult to get in and out. Fishing from the top of the bridges is possible but getting fish from the river to your hand proves difficult!
Two Bridges to Straight Cut
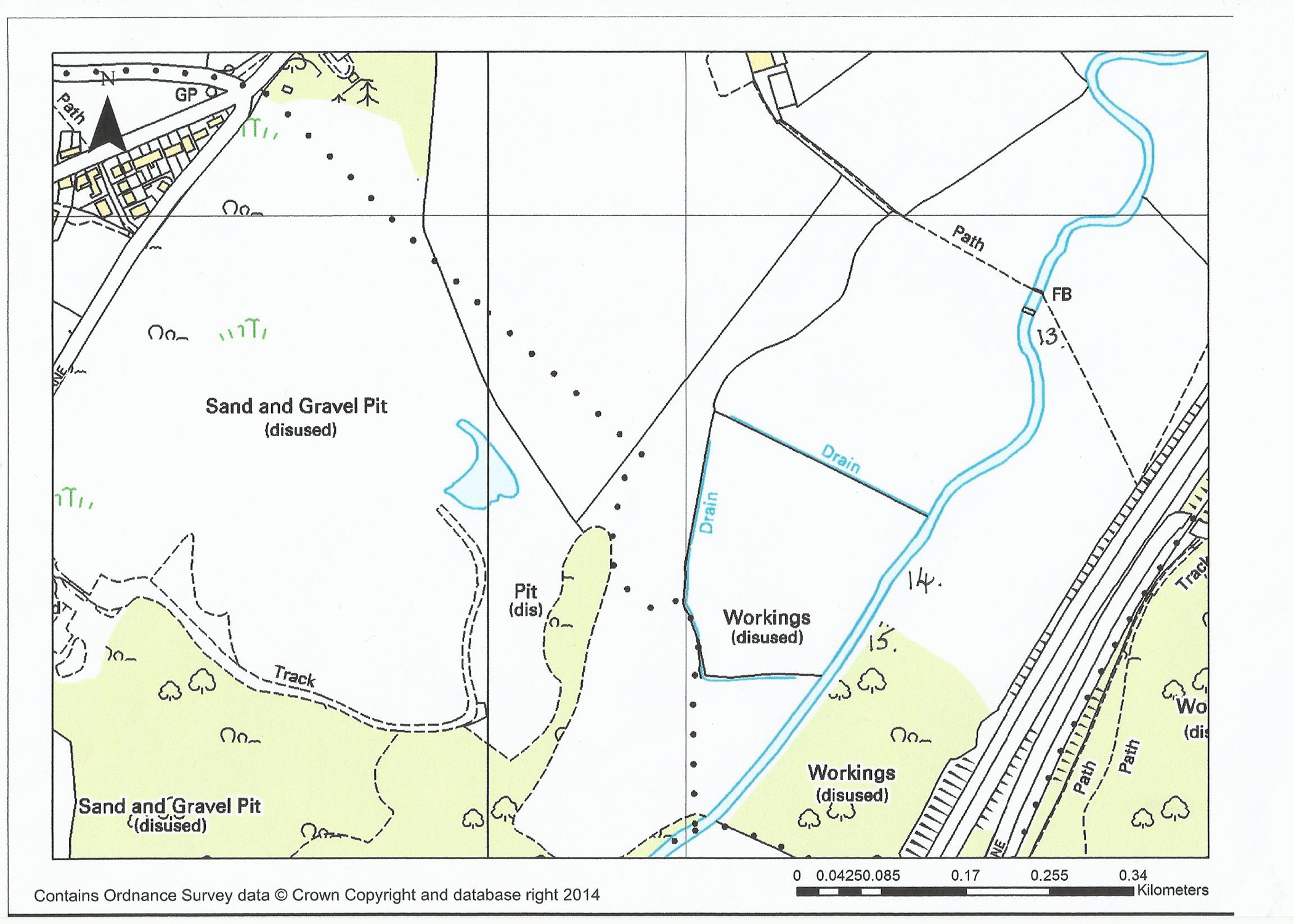
13. Just above the second bridge is a nice bend in the river with fast water flowing into a deeper pool. Fish can be found on the right-hand side as you look downstream.


14. Above this pool there is a shallow fast stretch that is quite wide, leading to a canopy of trees – all the way through to the straight cut. There are several deeper areas that are well worth exploring. In a couple of these areas there are regular rising fish. This area is a change from the willows of lower down as the trees are more natural and of a good size. There are quite a few entry points along this stretch ending with an interesting pool just before the gate to the straight cut.


15. We do not fish between the two gates of the straight cut, but have full access rights along the footpath.


Maize Field to Gresford Weir
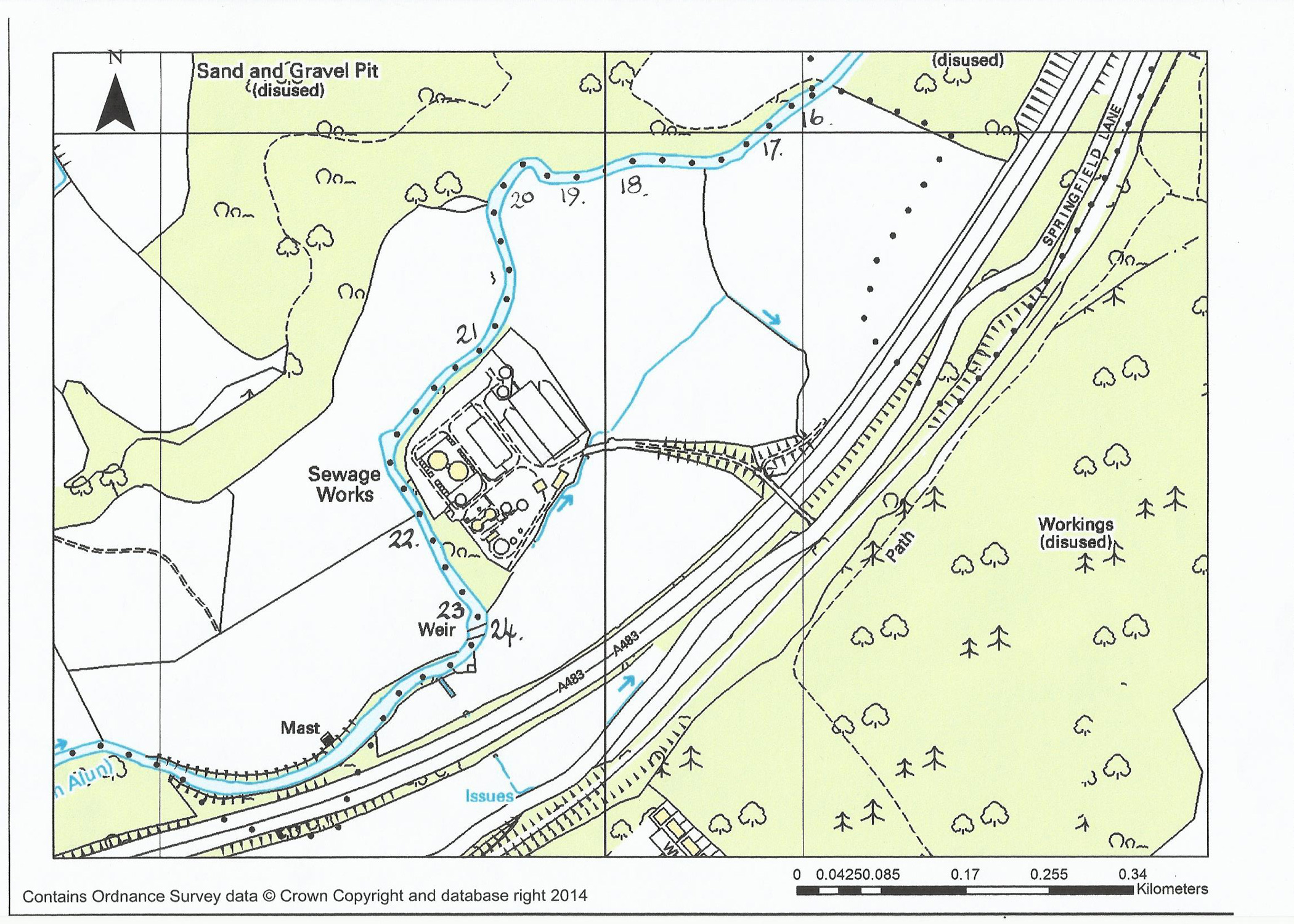
16. On entering the maze field there are three distinct pools along this stretch. Access to the first is just as you go past the gate and there is a short pool to where there are visible stones across the river in a ‘v’ formation creating some deeper fast water. If fishing this pool you will need to return to exit the river.


17. The next pool has steps down to just above the ‘v’ shaped stones. Once in the water here you can wade right through to kingfisher meadow on the left-hand bank, fishing as you go.


18. Kingfisher meadow is named after the colourful birds that you can often see here. There are several deep pools up to the stile by the sewage works. As you have a grass field behind you casting is theoretically easier! Free rising brown trout can be found all the way along this stretch. The difficulty is identifying what they are taking. In some of the pools drag is a major consideration.


19. There is a nice seat in the field here to have a rest for lunch or a cup of coffee.



20. At certain times of the year there are constant rising fish in this pool. Check what is in the surface film before selecting your fly as dead flies do seem to come out of the sewage farm with the clean water. The fish in this area can be put down with wading out but will return within 15 minutes. There are also large fish so beware. The largest one from this pool has been over 4lb.

21. The section between the sewage farm outlet pool and the weir is best classed as ‘pocket water’. There are a few deeper pools, one holds excellent grayling. As you move up to the island you will have a dilemma – if wading the top of the island it gives access to deep water, in front and to the right bank. The left-hand bank, however, has a channel that normally holds an odd rising fish, but it is extremely difficult to stop drag affecting your dry fly. As you pass the next pool you will find several ‘runs’ where odd fish can be picked up.


22. Just before the weir there is a deep slow pool that always holds fish.


23. Please remember the 30m rule as you approach the weir.


Gresford Weir to start of Worms Wood
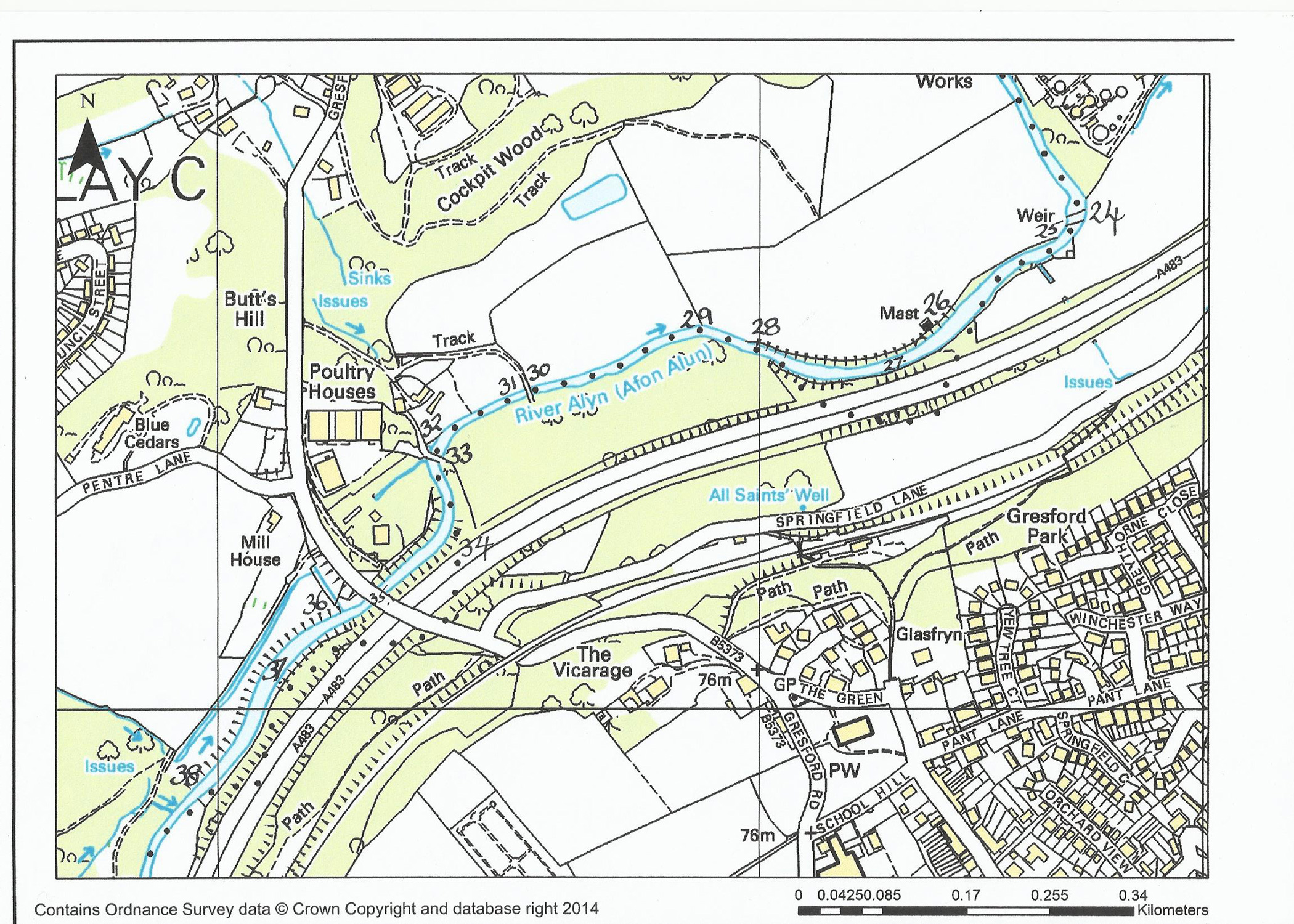
24. Upstream of the weir you are likely to see rising fish in the first pool. Although this is shallow it deepens towards the end of the 30-metre restricted zone. This area is wadeable starting on the left bank, but having to change to the right bank where it begins to get muddy. As you turn the corner you have access from both banks, but the better bank to fish from is the left-hand bank as you look downstream. There are several areas of deeper water and lots of shallow riffles that will create holding spots in warmer weather where the shallow water meets the deeper pools.


25.


26.


27. As you exit the wood on the right bank there is a pool immediately below you. When travelling downstream, if you get this far you have passed it! You need to enter the water from just inside the wood. From the fence line to the next tree is the deepest hole on this section. It is just about wadeable with chest waders in low water – be warned!! The left bank is the shallow area. Following this pool there is another pool on the left bank that has fast water coming into it but is not overly deep.


28. As you move upstream there are several nice pools on either side of the river and it is a matter of fishing out these pools.


29. This is the area below the farm bridge which has several interesting pools that fish hold in.


30. Upstream of the farm bridge looking towards September Cottage. Only one pool here at the moment as the rest of the water is thin and fast.


31. Drovers Bridge. Always worth a cast under the far bridge column.


32. This is upstream of the Drover’s Bridge which has a large deep pool on the left bank, but has a very tricky current to contend with. Normally both brown trout and grayling hold in this pool.

33. Area between Drover’s Bridge and the high road bridge from the Griffin Inn has odd pools in this length that hold a few fish, mostly shallow water.

34. Under the road bridge on the downstream side.

35. Upstream from the road bridge with the first pool on the horse field. Normally worth a fish for small brown trout, but can be difficult in low water.

36. The top end of the horse field where it joins with Worms Wood. Several pools along here – the right hand bank tends to hold the deeper water, but in low water conditions can be very difficult.

37. The last pool before Worms Wood – the trick here is to cast to the tree roots without catching them. Many flies have been lost here!! It normally holds a few good Grayling.

Start of Worms Wood
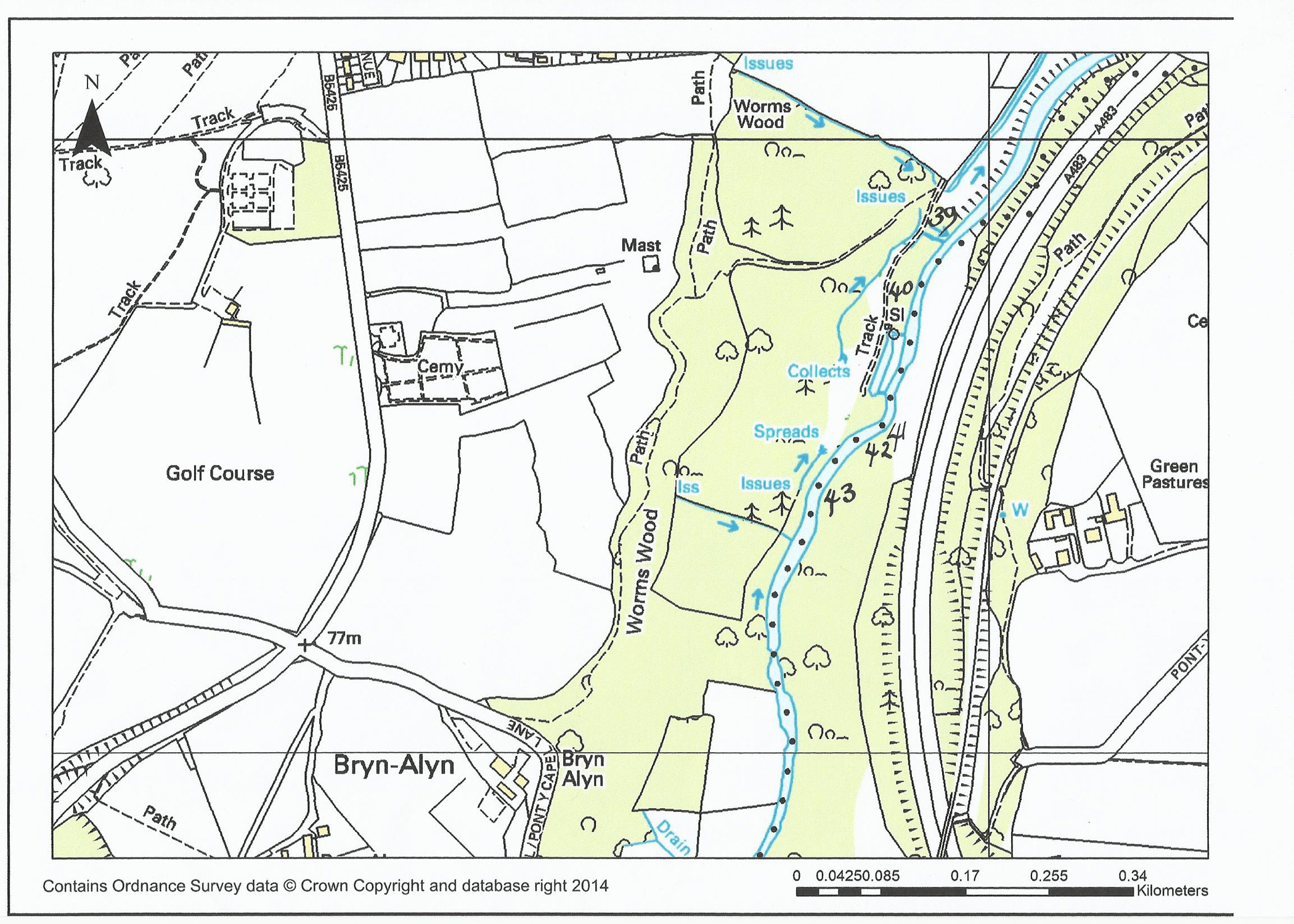
38. Entrance to Worms Wood. This area is mainly untamed and so offers more of a wild water experience. Paths are difficult to negotiate and banks can be steep at times and so access can be a problem. The initial path is on the left bank side, but you do have to cross to the right bank after the mill stones where the path continues to the end of our water. As you move into the wood there is a nice pool on the left bank where the deeper water is. This finishes where the tree lies in the water. The best fishing is under this tree.


39. As you move further into Worms Wood it is typical pocket water where odd fish can be extracted.


40. Crossing Point of river. As you follow the path along the left bank you suddenly appear at the river above the stones. This is a shallow water crossing (in low water with wellies). As you cross at this point you will see on the far bank a wonderful set of galvanised steps.


41. These galvanised steps lead from the dual carriageway down to the river. You can park at the top of these steps, which are located on the first lay-by from Wrexham to Rossett on the A483. Finding the path to these steps isn’t too hard, but getting back to your vehicle – just be aware that the first exit from the path does not lead you to the lay-by – it is 100 metres short of that along the dual carriageway. Best practice is to continue until you can see the junction of the horse field on your left and the lay-by should be just above you.


42. Moving upstream you will find that there are lots of obstacles in the river. There is very little maintenance to this section, due to its wild nature.


Middle Section of Worms Wood (End of fishing at Pont-y-Capel)
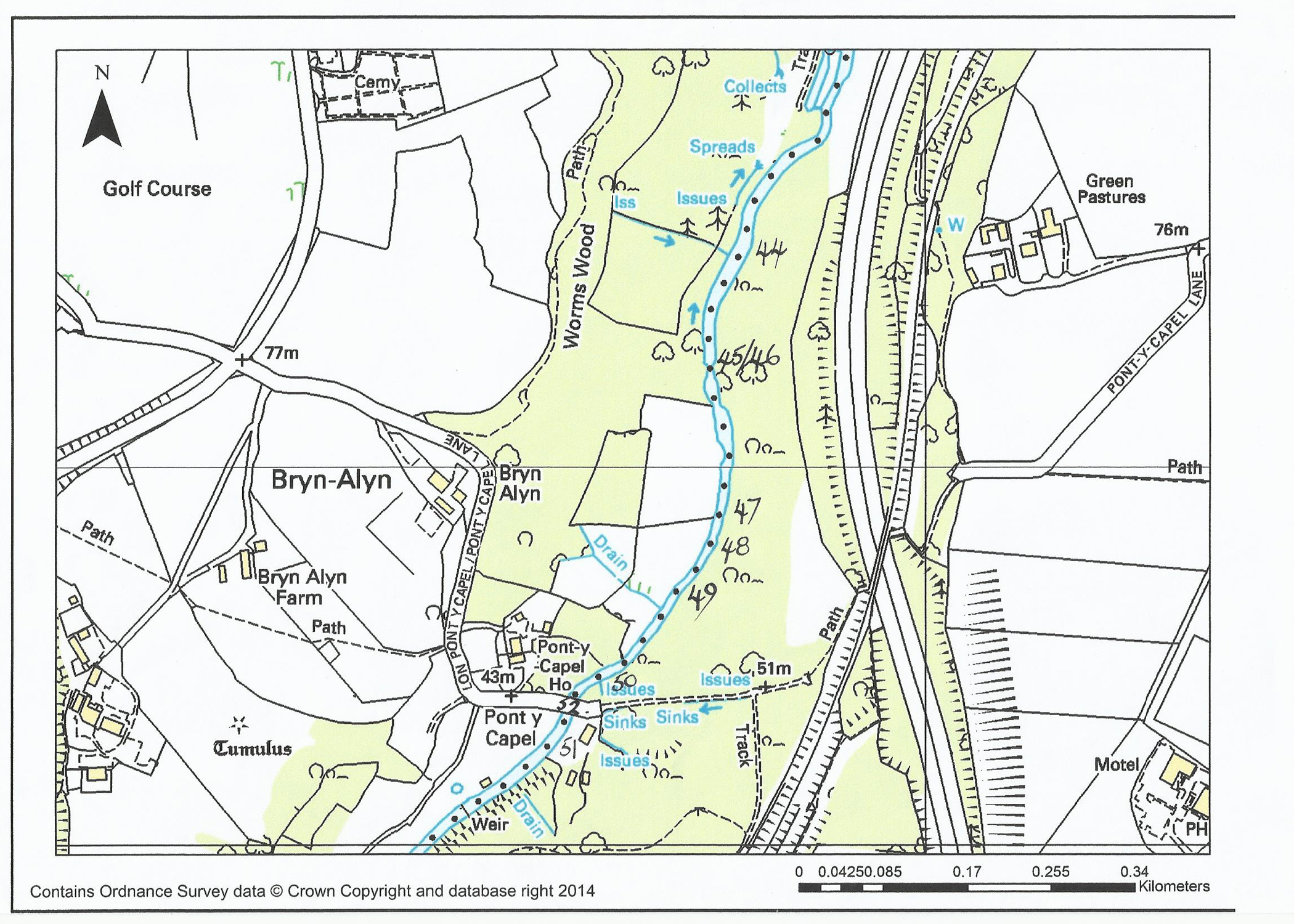
43. Moving further upstream again you will find several pools and runs leading up to a deep pool with rocks and chains on the right bank. Be aware that you may find the odd wild swimmer or two in here – it is very popular in the summer particularly with the local youths.


44.


45. Above the deep pool there is a shallow section followed by an old collapsed bank where work was carried out by the Council several years ago. The section above this is very productive for grayling. Fishing from the left-hand bank to the right hand bank there is a deep channel, so be aware of snags if fishing deep.


46. The first ‘obstruction’ – some may call it a weir? It has good deep water downstream of the concrete plinth, but is bypassed by the collapse of the river bank.


47. Upstream of the plinth there is a straight section that has rising fish on a regular basis, but due to the gradient of the bank and the location of trees casting is quite difficult.


48. Second ‘obstruction’ – For many years large grayling have been present in the deep water just below this obstruction.


49. We are now near to the end of our water at Pont y Capel. There are several pools and runs up to this point – brown trout make a good showing here, but can be difficult to tempt due to the slower pace of the river.


50. If you are fishing here you are now no longer in our water, so retreat back under the bridge!


51. Pont y Capel Bridge – this is the end of our water. Well done if you have made it this far!


Mavis Wilkinson.
Jul. 2017

That little bit of Difference
A dry fly that fools most fish, works well on streams, rivers and lakes. Too good to be true? What is it and where do I get them I hear you say. But before you rush off to tie some please read on, it is not without its problems. It can be a bit fiddly to tie, is not particularly robust and it is difficult to see at any distance when it is tied on small hooks. Still interested? What is it? My humble attempt at a Parraloop fly, with apologies to Ian Moutter. I have been using this pattern for over 10 years and it works well. Because of its small size fish take it casually, no real savage takes, just a drift up, thank you, and on with the snack. I say small size so I start at an 18 and get smaller, depending on the hook, I have gone as big as a 14, but found that it did not work as well. My favourite hook (if you can get them) is a TCM 101 in size 18 to 22. This is a straight eyed hook that is very strong. Alternatives are, Fulling Mill with an “Easy Eye” version which is the nearest, followed by Kamasan B440, which is a finer wire, Orvis 4641 Big Eye Dry Fly Hook and finally a Partridge 15BN Klinkhamer size 20 or smaller if you can get them.
There are just a few ingredients for this fly besides the hook and they are :-
Tail - Micofibbets (or cock hackle)
Body – Fly-Rite dubbin in Blue wing olive (or any dry fly dubbing of your choice)
Post (ish) – Black thread
Hackle – Genetic cock, Grizzle
Seems simple, not a lot to go wrong, but just in case a simple step by step guide :-
1. Mount hook in vice with a “Gallows” tool attached to the vice.

2.Next is to run thread down from the eye and bed in the tail (I normally put a wrap or two under the tail – with or without dubbing).

3. Attach a looped thread for the post and bed in well. (note for 3 & 4 this has to be tight as pressure will be applied)

4. Attach the hackle and bed in well.

5. Attach Gallows tool (if 3 is not well bedded in the thread will pull out, as thread needs to be tight)

6. Wind hackle up (distance is equal to hook length left to the eye ish), then continue to wind back down. Tie off the hackle at the base of the looped thread (at this point wet fingers and stroke back the hackle fibres so as not to trap them). Now the gallows tool can be removed, as the hackle will not unravel.

7. Run dubbing to the eye, leaving the hackle loose.

8. Now show the thread loop and hackle to the eye, if at this point it is too long, open the loop and pull gently this will shorten the hackle length. When correct, tie down, then reverse the thread loop and tie down again ? This stops the thread coming out too easily.

9. Cut off thread and smooth out hackles.


How to fish this fly
It depend where and when. If it is on a river then a single fly cast to a rising fish works for me. My best fish to this fly was a six and a half pound brown trout on the river Aire, below Malam Tarn. On lakes or small waters, I am tempted to attach a dropper tied New Zealand style if it is allowed. This dropper is usually a small buzzer type pattern(size 18 – 22) about 6” to 10” below, black is best, but a pattern to suit yourself. Again my best has been a Rainbow of over five pound on a 4 weight rod. Believe it or not, I find the hook hold to be far better with these small hooks and have rarely had a fish come off. In fact I struggle to remove the fly, even when using barbless or crushed barbs – this is when I can damage the Parraloop with forceps. What tippet to use, in my case 4lb, depends on the type and diameter of your normal line, but if you struggle threading through the eye with your normal brand, try the easy eye options as they have a much larger diameter eye.
Great fun. Go out and enjoy !
Eddie Wilkinson.
Jul. 2016

What do fish from the river Alyn eat?
The Alyn is the longest tributary of the Welsh Dee. The Alyn is quite different from the Dee, starts on limestone and has an alkaline pH of about 8.2. The river has an unusual invertebrate population and many club members have, for several years, assumed that the main invertebrate – gammarus pulex – forms a large part of the fish diet. Indeed the fish, both trout and grayling, respond well throughout most seasons to a pink shrimp pattern.
The Alyn’s link to the Welsh Dee is interesting because, some 40 years ago, an analysis of trout and grayling gut contents was carried out on the Welsh Dee by Dr John Woolland (1, 2). One of the surprises in his analysis was that terrestrials formed on average 25% of the diet of sampled fish, with fish from wooded areas eating far more. Surely on the Alyn, gammarus being so abundant by comparison with the Dee, terrestrials would not figure so prominently in the average diet?
How numerous are the gammarus? Well, Table 1 is based on the club’s Anglers Riverfly Monitoring Initiative (ARMI) data collected over several years. The data illustrates the scale of the gammarus population compared with other aquatic invertebrates such as baetis nymphs, caddis larvae etc. The figures given are the averages of several counts taken throughout the whole year, not just the season.
Table 1: Food item availability on the Alyn based on Riverfly ARMI data
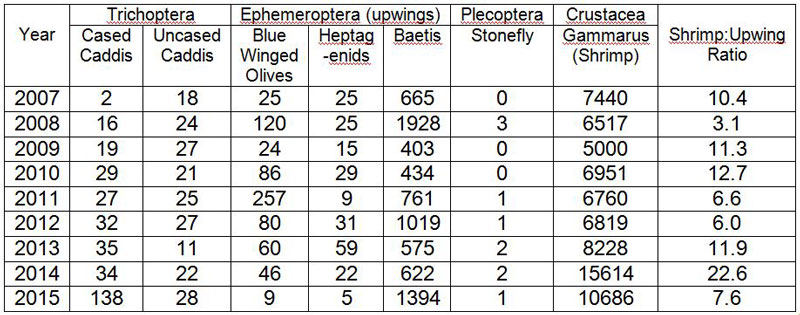
So, what are the fish eating? – gammarus, they are bound to be eating gammarus (shrimp) aren’t they? They are so nutritious and there are so many of them that they provide an easy feast for hungry fish, why would fish waste energy rising to catch flies on the surface?
Although there are upwing flies – baetis, blue-winged olives and large brook duns for example – their numbers are small by comparison with those of the gammarus. Hatches of upwings are not dense or frequent so fish don’t get time to key onto them. Steadily rising fish are seen rarely except when they are sipping hatching midge pupae – as a change from eating gammarus? However, it is almost always possible to find fish that rise fairly frequently, if not steadily, so dry fly fishing is often quite productive. So some fish eat things other than gammarus but, on the whole, most of us assumed that gammarus formed the mainstay of fish diets.
Assumptions are all very well but they don’t catch fish. During one RGFF committee meeting last autumn, the club chairman, Eddie Wilkinson, and I agreed to autopsy a sample of the fish that we caught throughout the season to see if we could confirm the assumptions, or find another explanation for the feeding behaviour of Alyn fish. We thought it would be useful to compare what the fish are actually eating with what we count as part of the Riverfly ARMI counts. We analysed the gut contents of both grayling and trout and the results for one short season are presented here.
We didn’t get started until late May but we then caught and analysed stomach contents throughout the season. This meant that we missed early fly hatches and any possible switch in feeding habits as the fish moved from winter through to spring and early summer. However, we have been able to draw some early conclusions.
The first trout, caught in May on a dry fly, contained mostly terrestrial diptera and blackfly (simulium) larvae whilst the first grayling had been eating the expected gammarus. The trout results were a bit of a puzzle but it was a stocked fish probably used to getting its food from the surface. However, it had found blackfly larvae attached to the riverbed stones or drifting in the current so we expected a gradual shift to gammarus as the season progressed and the fish got used to the full range of food available. The grayling, being largely a bottom feeder, had indeed been eating the expected gammarus.
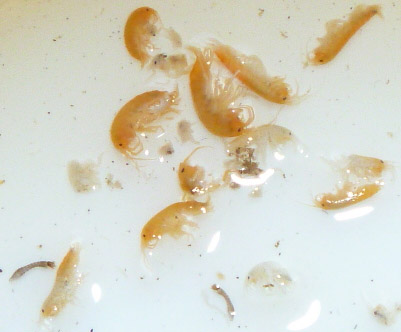
Contents of May Grayling
gammarus and simulium larvae. The gammarus look orange in this picture because the gut sample was stored in methanol before analysis. Methanol removes most of the pigment from the gammarus, leaving the orange colour.
The second and third trout, caught in June, added to the puzzle as neither contained any gammarus. The second, another stocked fish caught on a dry fly, had eaten a wide range of food items. Many were terrestrials – diptera and beetles – but it also had small caddis larvae and caddis pupal cases. In addition to showing a wide range of food items, the results for this fish showed that the stockies had rapidly learned to feed on natural items. The third trout was a wild fish caught on a nymph. It had eaten a bullhead, bloodworms and possibly a coryxa (or it may have been a beetle). In common with trout 1, neither of these fish had anything identifiable as either an upwing adult or nymph.

Contents of June trout
Snails, large and small beetles, small diptera, agapetes (caddis) larvae, caddis pupae cases.
The second grayling caught in July on a dry fly, had a couple of gammarus, small caddis larvae and baetis nymphs but mostly contained large and small diptera, a hoverfly, small beetles and midges. A more varied diet than the first grayling, probably indicative of more terrestrials being blown onto the water during what was a windy month.

Contents of July grayling
Hoverfly, large and small terrestrial diptera, small beetles, midges, gammarus, baetis nymphs, caddis larvae, simulium larvae.
Neither of the trout caught in August, the first on a nymph, the second on a dry, contained any gammarus. Both were wild fish. The first had been eating earwigs! The second had the most varied stomach contents yet - medium and small diptera, midges, numerous beetles from large to flea beetles, chironomid pupae, a small spider, a hoverfly, an ant, a caseless caddis and a caddis case. There were also remnants (legs, head etc.) of a very large diptera, possibly a bee.

Contents of August trout
medium and small diptera, midges, numerous beetles from large to flea beetles, chironomid pupae, a small spider, a hoverfly, an ant, a caseless caddis and a caddis case. There were also remnants (legs, head etc) of a very large diptera, possibly a bee.
We made a special effort in September. The crane flies should have been out in numbers and they often get blown onto the river. In addition, the Alyn has varieties that have aquatic larvae so the females fly over the water surface dipping as they lay eggs. Trout love a juicy crane fly so we expected to find them in autopsies. In addition, the female trout are developing eggs, for which they need carotenoids (the orange pigment) while the male trout are developing spawning colouration, for which they also need carotenoids. These pigments they get from their diet. The best source of carotenoids? Gammarus of course! So in September, we expected the trout to really switch on to eating shrimp!
What happened? The first grayling, caught on a weighted bug, contained very little. Just a few snails, small diptera, a chironomid pupa, a simulium larva and a baetis nymph – but no gammarus. The second grayling did have a few gammarus but had also been eating freshwater clams, snails and a cased caddis. The third grayling had developing eggs but had eaten only snails, beetles and bloodworm. The remaining fish were all trout, two of which were caught on dry fly. The first of these had eaten a wasp, nymphs (BWO ?), caddis (cases), snails and midge larvae. The second contained plenty of gammarus and snails – all indicative of bottom feeding, in spite of the fish being caught on a dry fly. The final trout, caught on a PTN was full of chironomid pupae AND large numbers of gammarus. None of the September fish had any evidence of crane fly, the only evidence being from an August fish that contained a pair of large wings that looked like those of a crane fly.
The season on the Alyn ends on 30th September for both trout and grayling, so further analysis of fish diets has not been possible.

Contents of early September grayling
Snails, diptera, chironomid pupa, simulium larva, baetis nymph.

Contents of late September grayling
Beetles, snails and bloodworm.
What does this tell us? That terrestrials, both diptera and beetles, form a large part of Alyn fish diets. The sizes of items eaten were very varied from midges up to what appear to be large beetles, a hoverfly and a bee. These were very opportunistic fish prepared to eat what the wind brought to them. Although caddis larvae, particularly the smaller ones such as Agapetus were eaten, as were pupal cases, we saw no adult caddis, even though two of the fish were caught on emerging caddis imitations. Until September, apart from the two grayling caught in June and July, not a single fish contained any gammarus. One of the grayling and two of the trout caught in September contained gammarus. The two trout in particular had been feeding heavily on them, especially the fish caught on a dry fly. So much for assumptions!
There are, of course, flaws in the analysis. Many of the fish were caught on dry fly and may simply not have devoted much effort to seeking out gammarus, although given the large numbers of gammarus available, that is unlikely. Several of the fish were stocked this year and may simply have been used to surface feeding. Again unlikely as they were well used to the river environment by the end of May and there was enough evidence that they were taking items from mid-stream and the bottom. The grayling did eat gammarus so they were available and one of the grayling had similar stomach contents to those of many of the stocked trout.
There is the possibility that the larger, wild, Alyn fish do eat gammarus but only one of these was caught. The trout caught by Eddie, mostly caught on nymphs, were larger than the stocked fish, and one had eaten a bullhead. The largest (4lb) and one of the others had been eating quantities of gammarus. These fish were full of eggs, as was the trout caught in late August.
All this gives us some suggestions but raises more questions.
1. Successful flies included whitehead PTNs, green and brown bugs, green paraloops, caddis emergers and Griffiths gnats.
2. We should try more artificals that imitate terrestrials – both flies and beetles.
3. If Alyn fish take such a varied diet of such differing sizes, they should be very tolerant of artificial fly size but small artificials seem to be better accepted. This is possibly because it is easier to tie a small rough, ‘buzzy’ fly than a larger close imitation.
4. Small caddis larvae are reasonably common and often drift downstream. Imitations of size 18 and smaller are worth a try.
5. The increased number of fish containing gammarus in September points to a seasonal change in diet, possibly to meet the needs of spawning.
6. We need to do more early season autopsies, not involving stocked fish, to check spring and early summer diets.
7. Most of the fish were caught morning or afternoon – before most adult caddis are about. Some evening-caught fish might contain undigested caddis.
8. Most of the fish were Y1+ or Y2+ (except for a 4lb trout). Next year we will include larger fish to see if/how diet changes with age.
The conclusions raise the question of the differences between the Riverfly counts and what we find fish are eating. The Riverfly count does act as a warning indicator of any water quality problems. It does not, however, necessarily indicate the full range of food available or what fish might be eating.
It is difficult to draw accurate comparisons with John Woolland’s findings of 40 years ago on a linked, but different, river. It is, however, possible to try to do some comparisons however by looking at the Riverfly ARMI counts and the table of food items in Woolland’s final article (2).
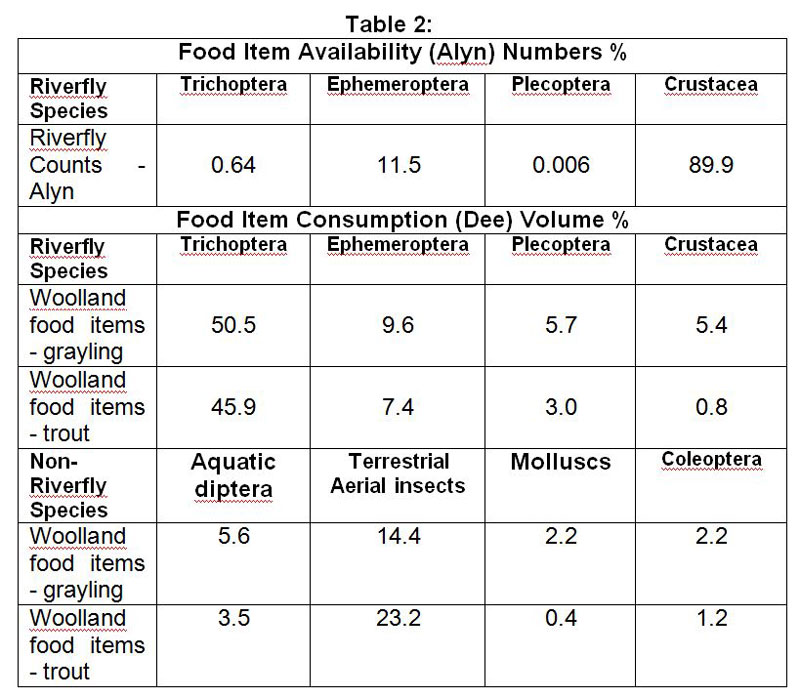
To get the Table 2 figures, some figures have been bulked together, such as Trichoptera, Ephemeroptera and Plecoptera larvae/nymphs and adults from Woolland’s data. We do not yet have food items numbers for the Alyn so the table is trying to compare numbers (%) with Woolland’s volume (%). The availability numbers add up to 100% but the Woolland numbers do not as they leave out items not measured in Riverfly counts such as terrestrials, most of which have been included in the lower part of the table.
It is interesting that so few crustaceans (mostly gammarus) were also eaten by Dee fish, although the Dee is noted for its high caddis populations. It is also interesting that the Alyn autopsy findings showed coleoptera (beetles) in most samples whereas the Dee fish had eaten very few. All of the groups in the lower part of the table were present as important diet items in the Alyn fish (Table 3). As far as the fish are concerned, what the Riverfly counts leave out is at least as important as what the counts include.
The main question remains - when Alyn fish seem so ready to take a pink shrimp why are there so few gammarus in their diet? This question is, to me, particularly pertinent as, in alkaline chalk streams, gammarus can form over 90% of grayling diets (1). John Woolland concludes his Spring 1987 article by saying “Grayling generally fed on organisms which were most numerous or most accessible”. Alyn gammarus are certainly numerous, seem no less accessible than in other rivers but are not consumed in the numbers assumed – why? Until we catch a talking fish, we can only guess!
Acknowledgements.
Thanks to Howard Stevens for comment on the draft manuscript, and to the RGFF committee for encouraging the work.
References.
1. Woolland J, (1987), Grayling in the Welsh Dee, part 3, Feeding. Journal of the Grayling Society, Spring 1987.
2. Woolland J, (1988), The feeding relationships of grayling & trout on the Welsh Dee. Journal of the Grayling Society, Spring 1988.
Les Jervis and Eddie Wilkinson.
Oct. 2015

Ode to a New Season.
(with apologies to the Original and Well-Known Rhyme by Anonymous, Ogden Nash and many others)
The spring is sprung, the grass is riz,
I wonder where those fishes is.
They say the fish is on the fin,
But that’s just tish, the little fin is on the fish.
Now April’s here, blackflies about,
But flies to use have me in doubt,
If only LDOs did hatch
I’m sure I’d find a better match,
The trout to catch,
When all else fails.
May at last, the flies are out,
The fish are smutting,
Klinks no doubt will catch those fish
And pull ‘em out.
Back home in June the weeds do sprout.
A deadly duel now ensues,
(for most of you that won’t be news).
“Going fishing – in your dreams,
Cut the grass and dig the ground,
For Sunday lunch, my mother’s round”
I sweat and toil, the day is long,
My mind on other things belongs.
If I can get away by seven,
I’ll get a couple of hours of heaven,
Wrapped in the fins of my sweet love.
The trout await, I cannot linger,
Then secateurs cut through my finger.
Blood pours out, the wife tut tuts,
And lets me off the garden cuts!
“Cover that with gauze and gum –
Then bugger off and have some fun”.
September’s here, the weather’s dank,
The weeds are dying on the bank.
Those grayling are no fishy fools,
They’re swimming off to deeper pools.
I’ll need to use some Czechs of leads,
And hope it lands upon their heads.
At least that should make ‘em think,
“Ahh, here is something Eddie pink,
I’ll grab it now before it goes, and flies away”.
In just two days the season ends,
My fish ambition still suspends.
A final trip, my PB fish?
Ah well, I can always wish!
Les Jervis.
Apr. 2015

Fishing in Cape Verde.
In February we had planned to go Skiing, but when we tried to book a price of £4,000 each was the best we could get (without ski passes or ski’s)! So a new plan was needed. As we have been to most “local” winter sun spots, a new venue was needed. Cape Verde is a range of islands off Africa, about 2 hours flying past the Canaries.


I did check out the fishing and found there was plenty availability, but not fly, as there could be a problem with waves. I thought I could get round this (maybe). The species on the cards were shark. I had a couple of evenings for just an hour at a time (light goes very quickly). The first time I had two fish on, but they quickly cut through the line, must remember a wire trace next time. Also what can happen when “friends” drop by for dinner (see later). As the waves were “interesting” I did use a fast sink line to get below and into what I thought was the feeding zone.


Following my steep learning curve, I changed to a dry line, to get away from the sharks. I pointed out to my wife where the “other” fish were – in the wave, they could be clearly seen swimming in the “curtain”! It is the 1st time I have had to cast uphill to fish, what a novelty. There was however a glitch. I managed to hook two more fish, but, once they were hooked they ran with the wave until it crashed – the fish were literally ripped off the hook.
There was another issue though, when a fish was hooked, the “others” did try to take advantage. When one of the small shark about 3’ long, but quite thin, was hooked and on its way in, another larger shark decided to join in and have dinner on me, the fish was literally bitten in half. Oops, not sure I would like whatever took the other half on my 7 weight travel rod , well actually yes I would, please!!

Final plan was to move a few miles up the beach to a calmer area in the lee of an island, where there were plenty of fish and coral. Only found out about this area late in the holiday and by then there was however a slight problem, some fool decided to “play” in the waves and came off second best. I came down on my head and neck, causing a lack of use in my left side. Yes I am in the picture below!

Not good when trying to fish, so another trip to be planned for the near future, provided the arm recovers. For anyone thinking of going, we stayed on the Island of Boa Vista, with a population of 6,000, all very friendly. The hotel was fantastic, not our usual choice, too big, but it was recommended to us . It was all inclusive, and it really was, no limits.


Yes it did look like a palace. There is only a clinic on the Island and a small police force. If you go into the local village, you will get some attention, but no extreme hassle. The locals tend to leave you alone if you tell them. It is however a very poor area and locals will try to get you to buy “local” crafts. This is an up and coming tourist area that will catch up with the likes of the Greek Islands or Egypt in the near future – Get in quick, but remember the rod.
This is the fly that worked: -

Eddie Wilkinson.
Mar. 2015

The OTHER Blackflies.
The most recent Anglers’ Riverfly Monitoring Initiative (ARMI) bug count, in February, showed a significant reduction in gammarus and an increase in upwing nymphs, mostly baetis , in most of the sampling areas. What was also seen, but not counted for ARMI, was a big increase in simulium blackfly larvae and pupae. These flies have increased dramatically in the last couple of years and I recently gave some links to possible imitations.
The simulium increase has taken place against a background of other blackfly on the river. These, I think, are mostly non-biting midge of the chironomid type. These often occur in huge numbers and their larvae (bloodworms) and pupae are important sources of fish food. The club Chairman, Eddie Wilkinson, sent me a photograph of the stomach contents of a trout caught in the sewage pool. It was crammed full of chironomid pupae.

It seem sensible to have a few chironomid imitations in the fly box as these are food items available to trout and grayling all year round. Imitations of pupae– the stillwater buzzers – are probably the artificial of choice and are easy to tie. However, bloodworm can be effective, especially if fished deep in slower parts of the river.
There are many more chironomid imitations than those available to match simulids. River chironomids are often smaller than those from stillwaters, so stepping down to 18 – 20 hooks, smaller if you can manage, is a good idea, with size 16 being a big fly. Colours can vary widely but pupae imitations fished in the top couple of feet of water should have some flash and a bit of claret/red built in. Some anglers do well with Shipman’s buzzers but they have never worked for me, a smallish version of the Eikre pupa gives me more confidence - and fish. Below are some photos and links to possible flies and tying instructions. Mostly these are US or Australian/New Zealand patterns but I’ve found that they work well. The Eikre pupa is a Swedish fly and tied small (I use a TMC103BL size 17), works well. If all else fails, I find the Griffiths gnat a reliable backstop. Tight lines!
There is a good fly fisher’s summary of chironomids at: http://www.flycraftangling.com/index.asp?p=125.

Emerging adult: Eikre pupa
http://shop.siman.cz/index.html?pod=/cl1801735980.htm.

Black Smut
http://troutterritory.com.au/fishingintasmania/trout-flies/black-smut/

Yamamoto's Foam Wing Midge Emerger Type 1
http://www.flyanglersonline.com/articles/eotg/2014/eotg20140922_SatoshiYamamoto.php

Yamamoto's Foam Wing Midge Emerger Type 2
http://www.flyanglersonline.com/articles/eotg/2014/eotg20140922_SatoshiYamamoto.php

Bulbous Gnat Emerger
http://gwilymhughes.com/THE_GH_BULBOUS_GNAT.asp

Shaving Brush / Black Funnel Emerger
http://globalflyfisher.com/patterns/black_funnel/

“Too simple bloodworm”
http://flyanglersonline.com/alcampbell/ac012604.php

The Bloodworm
http://www.graylingsociety.net/docs/grayling-news-49.pdf NB The dressing appears at the bottom of this page.
Les Jervis.
Mar. 2015

Opening Day 3rd March '15.
Graham had still not forgiven me for planning a trip instead of going fishing on the last day of the season, so I could not miss the beginning of this season could I? As per last year we decided to fish the top of our river, starting at the horse field. Graham had decided to drive, as he struggles with getting in my Mazda MX5, and so he picked me up at 06:15am! The river had been up and coloured for a good few days, so much so that the river working party on Sunday had to stay on the banks due to high fast water. But by Tuesday the level at Pont y Capel had dropped to just over 0.4m, so just fishable, but it was going to be hard. We fished out the horse field with Graham suggesting that I fish first so that he could understand my methods. So much for that, I failed to get a touch. Moving into Worms Wood Graham showed his true colours and had the 1st fish, a nice little Grayling.


As can be seen from the photos it was cold, but we had expected that, it just seemed a little slow, maybe it was just too early for the fish? My first fish was this monster of a brown trout. As we moved on the fish started to wake up and including a couple of nice grayling, the best being 39cm plus long.


Access at times was difficult, due to deep water, the sudden drop off’s that weren't there last year, “new” trees and obstructions plus the very fast runs, that at times were too fast! If you are contemplating a trip please take care.


The catch returns for our first day: -
Graham - 9 Grayling (best 3/4lb)and 3 Browns (best 1lb+) total 12 fish
Eddie - 16 Grayling (best 11/2lb) and 5 Browns (best 3/4lb) total 21 fish



The flies used are all variants of those tied at our Fly Tying Evening held on the 1st December and their dressings are given in the article below. The variation being all the flies have a back of mirror flash.
Eddie Wilkinson.
Mar. 2015

Blackfly artificials - A challenge for 2015.
The Alyn fly life has been monitored for several years under the Riverfly Partnership Anglers’ Monitoring Initiative. The volunteers who do the bug counting have often expressed concern over the very large numbers of Gammarus, and the low numbers of upwing nymphs. Club members know that the fish do not rise readily and fly hatches are rarely seen. In recent years, bug counts have identified increasing numbers of blackfly (simulium) larvae and pupae in the Alyn (see “Fly Life on the Alyn” under the Articles tab). Recently (Feb 2015) Howard Stevens and I were collecting some gammarus samples and were amazed at the increase in blackfly larvae and pupae. Some, but not all, volunteers have found the same. In some parts of the river, gammarus still far outnumber everything else, yet just below the sewage treatment works, the most recent count showed lower numbers of gammarus and healthy numbers of baetis nymphs. A truly confusing picture of a changing river. As a single female adult simulium fly can lay up to 200 eggs several times in her lifetime, the rate of increase in numbers can be phenomenal and blackfly can become a major food item for trout and grayling. I’ve included some additional photos of blackfly at various stages in their life cycle. The photos are from Dwight Kuhn’s excellent website, http://www.kuhnphoto.net/.
It seems very likely that large numbers of simulium blackfly are here for at least a while. Trying a blackfly artificial is a good bet when fish are sipping down something small. The simulium prefer fast water so try a riffle.
Although I gave a couple of patterns I thought might be suitable imitations (see article under Article tab, “Fly life on the Alyn – Post Script”) and which work well for me, some recent internet browsing has produced a number of patterns that look well worth trying. The patterns are from experienced tiers who recognize the importance of blackfly as trout food items.
The larvae, which are filter-feeders, living on bacteria, algae and suspended organic material, are often exposed on stone surfaces or vegetation and are easy grazing prey. The pupae are usually under stones or on woody structures so are probably less-easily eaten by trout and grayling. The adult fly emerges from the pupa fully-formed, encased in air and rapidly rises to the surface. This makes an inviting silvery target, easily preyed on by fish. The adult only stays on the surface for a very short time and the female returns to lay eggs on or under the water surface, attaching the eggs to branches, grasses etc. trailing in the water.
If you fancy a bit of a challenge, and can tie small patterns, these are well worth trying during the 2015 season. You might also want to try your own ideas for an artificial blackfly. I have a number of ideas that I want to try, and remember, blackfly form a significant part of the trout diet and their numbers are increasing dramatically in the Alyn. Good luck and tight lines! The Jeff Morgan “west fly” article, part II, gives both patterns and recommendations for fishing.

Simulium larva:
Prefer faster water in riffles. A possible target for artificials and a couple of patterns are suggested in the website. Remember that the larva has a “coke-bottle” shaped body.

The simulium pupae:
The pupae are often under stones and may be a difficult stage to mimic.

The emerging adult:
Looking rather like an Alien, an emerging, fully-developed, adult with an air bubble. Makes a fast trip to the surface but, en route, it must be an attractive target. A small black artificial with plenty of silver/flash might bring results.

The adult on the surface:
The fully developed adult on the surface, but not for long! A small black midge imitation should work well. Something like an American Trico dry pattern?

Female adult feeding on a human arm:
I don’t know if the simulium living in the Alyn have adult females that can feed on human blood. I’ve not been bitten – yet. The females need a blood meal for egg development, so watch out, the bites can be painful!
Useful Websites
Herbst, Ed. Backfly – the challenge: -
http://www.tomsutcliffe.co.za/fly-fishing/friend-s-articles/item/957-black-fly-–-the-challenge-by-ed-herbst.html
Morgan, Jeff – Black flies, Part I (background): -
http://www.west-fly-fishing.com/feature-article/0204/feature_657.php
Morgan, Jeff - Black flies, Part II (patterns): -
http://www.west-fly-fishing.com/feature-article/0204/feature_658.php
Jeff Morgan – Fly Tier: -
https://planettrout.wordpress.com/tag/jeff-morgan-fly-tier/
Al Miller – Tying Al’s Trico: -
http://howtoflyfish.orvis.com/fly-tying-videos/dry-flies/741-als-trico,
Dwight Kuhn – photographer: -
http://www.kuhnphoto.net/.
Les Jervis
Mar. 2015

Simple Sinkant.
If you have not tried this before why not give it a go, it is remarkably simple to do and cheaper than the commercial options. It means that you can keep some in each fishing jacket, bag or creel and never having to think "I should have put that one tub of sinkant in my pocket before I left home".

Here are the bits and pieces you will need: -
- Fullers earth – a quick search on the internet found the bag in the picture for £1-40p including post and packaging.
- Glycerin – from your local Boots
- Washing up liquid – from your kitchen
- Xing – (optional) a couple of drops – found in the bottom of a fishing jacket
- Mixing bowl – sized to suit your needs
- Pots to put it in after mixing – See later
- Tea spoon, spatular or knife
Start with a couple of tea spoons of Fullers earth, then add the Glycerin, plus a dab of washing up liquid. Finally add a drop or two of xink (optional) or even some scouring powder to removes glare off a floating line if you like.



Mix to the consistency you require, some like it fairly stiff like putty, others prefer it softer like tooth paste. The consistency you choose will influence the container now needed. For those who chose the stiffer mix remember 35mm film containers, these are ideal for a full season (or more). Just fill it up and pop the top on, it will lasts for years.

If you go for a thinner mix then another option could be “Hotel mini shower bottles”. I am sure no one takes these from hotels, mine were “found” in a case on returning home, must of fell in.



Unscrew the top and open up the small hole to the full size of the threaded bit with a sharp pen knife. Then feed in your mix with an appropriate tool, mine is for scooping out lobster meat. Put the top back on and you then have a squeeze supply of sinkant that does not take up much room or leak.
All ready for the new season (ish)
Eddie Wilkinson
Feb. 2015

Patterns from the Fly Tying Evening.
These are the patterns from the Flying Tying Evening held at the Griffin Inn on the 1st of December. The event was lead by Eddie Wilkinson, you might recognize the patterns as those handed out by Eddie at the River Walk last May.
Pheasant Tail Nymph

| Hook: | Kamasan B175. |
|---|---|
| Tail: | Pheasant tail. |
| Body: | Pheasant tail. |
| Rib: | Fine Wire. |
| Collar: | Micro synergy straggle fritz - gold/black. |
| Hackle: | Greenwells. |
| Head: | Tungsten Bead. |

This variant used goose biot for the tail and for 'aiming point cheeks' and the collar has been omitted.
Green Parachute

| Hook: | 16 - 20 TCM 101. |
|---|---|
| Thread: | Green Ultra 70. |
| Tail: | Ginger cock. |
| Body: | Green Fly Rite /poly dub. |
| Post: | Hi Vis Green Antron. |
| Hackle: | Grizzle cock. |
Pink Shrimp

| Hook: | Kamasan Grub 10. |
|---|---|
| Thread: | Green. |
| Under Body: | Lead wire. |
| Body: | Micro synergy straggle Fritz - Pink. |
| Rib: | Fine gold wire. |
| Back: | Plastic Brown Raffia. |
General Midge pattern

| Hook: | 18 - 26 TCM 101. |
|---|---|
| Thread: | White Ultra 70. |
| Tail: | Krystal Flash UV Pearl. |
| Body: | as Thread. |
| Hackle: | White palmered. |
Eddie Wilkinson
Dec. 2014

Fly Life on the Alyn - Post Script.
Following his article 'Fly Life on the Alyn' Les has written this article listing the flies he uses. Not only has he supplied the pattern dressing but has included links to various youtube links so that you can watch the fly being tied.
Tying a small Klinkhamer
www.youtube.com/watch?v=t-as9IV1-d8
Not really a Klinkhamer – a small parachute emerger but it works! I tie it in black with a grizzle hackle. I sometimes rib the body with fine lurex. This tends to work well on cloudy days and in the evening.

| Hook: | Partridge Kinkhamer extreme, 18 – 20 or suitable emerger hook works just as well. |
|---|---|
| Wing: | Poly yarn - white. |
| Body: | Fly Rite Black Poly Yarn. |
| Hackle: | Grizzle cock. |
| Thread: | Fine black. |
Tying a SupaPupa
http://ukflydressing.proboards.com/thread/2088
A good description of an excellent fly. An alternative dressing is given at:
www.youtube.com/watch?v=fJ5li1kiZ4Q
In this video, Davie McPhail uses a CDC wing to make the fly more visible. I use a white poly yarn or aero-wing post and, again, a grizzle hackle. Don’t make the flue too long, it interferes with hooking. But not too small as the fly is much less attractive. The critical bit is to cut off the hackle fibres above and below the hook to give a fly that lies in the surface film.

| Hook: | Tiemco 103BL, 15, 17, 19. |
|---|---|
| Body: | Fly Rite Poly Yarn, cream for rear 2/3 and brown for front end. Almost any contrasting colour variation works well. |
| Hackle: | Grizzle or red game. |
| Thread: | Fine light olive. |
Tying an Orange and Partridge Spider
www.youtube.com/watch?v=iuK61AFV0NU
| Hook: | Down eye wet or spider, 14 or 16. |
|---|---|
| Body: | Orange tying silk. |
| Hackle: | English grey partridge back feather or Grizzle hen. |
I do several variations. An orange thread body fishes just under the surface. Using orange ultrawire fishes deeper and putting a small tungsten bead after the eye fishes deep. You’ve probably guessed that I use a grizzle hen hackle so I suppose the fly should be an orange a grizzle spider. Fish don’t seem to mind!
Tying Goddard's Black Gnat and Smut
I couldn’t find videos of tying for either the Black Gnat of the Smut but the are very simple. Don’t make the wing too large on the gnat, it is just there to act as a trigger and too much can startle the fish rather than attract them.

Goddard's Black Gnat
| Hook: | Wide gape upeyed, 14 to 22. |
|---|---|
| Wing: | small bunch of pearl lurex. |
| Body: | Black Poly Yarn. |
| Hackle: | Three turns of black cock. |
| Thread: | Fine black. |

Goddard's Smut
| Hook: | Fine dry 18 to 22. |
|---|---|
| Body: | Three turns black ostrich herl. |
| Hackle: | Two turns of black cock. |
| Thread: | Fine black. |
Tying Goddard's Suspender Pupa
www.youtube.com/watch?v=a3qeZkCH7tE
In this video, John Smith ties an easier variant.
Instead of fixing the polystyrene/ethafoam ball in fine mesh from a lady’s stocking, I thread the ball on to 4x leader material, wrap the material to the hook shank before tying the fly, then finish off by carefully melting the leader material so that it forms a blob that stops the bead coming off. Not as robust as Goddard’s method but works well enough and solves the problem of getting the mesh material!
| Hook: | Drennan emerger or similar, 16 to 20. |
|---|---|
| Thread: | Fine in same colour as body. |
| Thorax: | Peacock herl. |
| Body: | Antron body wool. |
| Hackle: | Grizzle cock. |
| Tail: | Small bunch of white nylon filaments 1/4 of body length. |
Tying the Griffith's Gnat
www.diptera.co.uk/patterns/g/griffiths_gnat_ti.htmlOr:
www.intheriffle.com/fishing-videos/fly-tying/griffiths-gnat/

| Hook: | Standard dry-fly hook, 14 to 24. |
|---|---|
| Thread: | Black, 70 denier or 8/0. |
| Body: | Peacock herl. |
| Hackle: | Grizzly hackle. |
| Head: | Black tying thread. |
I tend to use a larger hook and only dress the first half to two thirds as I struggle with small hooks. The larger hook does not seem to put off the fish and I think gives a better hook-up rate. If you prefer a bigger hook, try tying a Double G, same materials. Both work equally well. For smaller flies I tie in a white poly yarn post for visibility. With all my flies tied on smaller hooks, I follow John Goddard’s advice of slightly offsetting the hook point for improved hooking.
Double G

Now that the close season is upon us, tying time is here. If you do decide to try any of the above, I hope they work for you. Some, like the Klink, the Griffith’s Gnat, Double G and the SupaPupa work for me all through the season. The spider works well during the early part, March to June. The Goddard flies I keep for the later season wary fish. Tight lines in 2015!
Les Jervis
Nov. 2014

Fly Life on the Alyn.
Is there a problem with fly life on the Alyn? The simple answer is probably. The real answer is much more complex. Fly life certainly seems to be changing and, with it, feeding habits of the Alyn fish.
The Alyn is a post-industrial river and has been dramatically cleaned up in recent years. With the clean-up have come changes in the life the river supports. This applies to the increase in the grayling population, the reappearance of otters and changes to the invertebrates, including aquatic flies that form part of the food chain of the fish.
Clean-up and some on-going problems
However, there have been some negative changes to water quality as well as the positive effects from the post-industrial clean-up. Changes in land use have probably led to increased run-off containing pesticides, herbicides and fertilizers such as phosphate and nitrate. Increased volumes of waste-water from sewage plants are a problem because, although the treated water is cleaner, it contains many new chemicals – antibiotics, oral contraceptives, analgesics etc, many of which have been shown to affect fish and invertebrates adversely. Changes in rainfall pattern also affect the river with periods of low rainfall increasing the concentration of any deleterious chemicals from run-off and waste-water inflow. Sudden high rainfall has caused much damage, generating more work for the volunteer working parties and destroying much of their remedial work to the river and its banks. Invasive plant species like Himalayan balsam cause huge problems, not only of eliminating native plant species, but also de-stabilizing riverbanks and vastly increasing erosion in winter. The increased sedimentation chokes the river-bed, redds, weedbeds and fly larvae causing at least as much damage as any of the other changes mentioned.
Paul Leng’s excellent little book (ref. 1) makes very interesting reading and gives much information on the ever-changing Alyn. It certainly gives a good background to the current perceived problem of fly life.
Gammarus
Gammarus populations in the RGFFC waters are exceptionally high and are seen by many as the main reason why fish do not rise freely, either to hatches of natural fly or to the well-presented dry fly. With so many gammarus freely available, why would fish bother to waste energy rising to surface flies? In addition, although gammarus mainly feed on rotting vegetation, they are opportunistic predators and will catch and eat fly larvae (and each other) and will decrease fly numbers. So, are gammarus the main reason why fish are not rising to feed? Live gammarus samples taken from the Alyn recently were tested for predatory behaviour in the laboratory of Professor Jaimie Dick of Queen’s University, Belfast, by his postgraduate student Helene Bovy. She found that, in common with other mainland UK gammarus, Alyn gammarus were smaller than those from Northern Ireland and ate fewer prey species (Baetis nymphs). Several graphs from Helene’s data are shown (Figure 3).
Whilst the Alyn and Galmington Stream gammarus are essentially identical, the Alyn gammarus eat fewer prey than those from any of the Northern Ireland streams. This does not, of course, mean that gammarus predation on upwing fly larvae is not a problem on the Alyn – the numbers are so large that they well compensate for any lower individual level of predation. However, taken together with the Riverfly sampling data that shows upwing larvae even in the presence of high gammarus numbers, it suggests that our gammarus concentrate their feeding mostly on rotting vegetable material. One of the problems about gammarus is that they are prolific breeders, often mating several times a year. As a result they are very quick to colonise any areas where fly life has been wiped out by pollution or is at a low level. I suspect that we have so many gammarus because they are taking advantage of available niches not already occupied by fly nymphs – the lack of fly almost certainly preceded the gammarus. The conditions in the Alyn are ideal for them in terms of pH and the amount of available vegetable matter from fallen leaves so their numbers have exploded.
Riverfly Sampling Data
The RGFFC waters are very fortunate in having several years of fly life monitoring under the Riverfly sampling scheme so the accumulated data is probably as good a place as any to start looking for patterns, answers and possible solutions. The data certainly highlights the very large numbers of gammarus. Table 1 summarises the data for gammarus and upwing fly larvae since 2007. From the data it appears that although numbers of both gammarus and upwing larvae are variable, the relationship between the two is not simple. Higher gammarus numbers do not automatically mean a lower upwing larvae number and vice versa. What the numbers might mean is that in years of high gammarus numbers, fish find it more difficult to locate the upwing larvae and concentrate their sub-surface feeding effort on gammarus. That does not explain why, when upwing larvae are hatching, the fish still tend to feed sub-surface.
Autopsies
It is very tempting to assume that fish are locked on to feeding on gammarus, but what evidence do we really have? Figures 1 and 2 are from autopsies of freshly caught fish and suggest otherwise. Although from different species and different sections of the Alyn, neither autopsy shows any evidence of gammarus, in spite of their overwhelming predominance in invertebrate sampling. Unfortunately, these are the only photographs of stomach content I have but they do highlight that, in the sewage pool trout, chironomids are the main food item whilst in the lower Alyn grayling, adult flies and gnats are major elements, along with bloodworm and unidentified bits detritus. Again, there is not enough data here to draw conclusions other than we cannot make assumptions about what fish are eating and we need more autopsy data throughout the season.
What we don’t count and artificials
One of the deficits of the Riverfly sampling data is that what is NOT recorded is at least as important as what IS recorded. Caddis, upwings, stonefly and gammarus are counted but not chironomids, bloodworm, midge/mosqito larvae, leeches, snails – all valuable food sources for fish. Amongst the midges, the blackfly larvae and pupae (Simuliidae – reed smuts) were present in significant numbers in the samples taken in September 2014 by Mark Pierce and me, just downstream of the sewage pool (Figures 4, and 5). The species of blackfly has not yet been identified but if anyone has been bitten by swarms of blackfly recently, please let me know (e-mail: lesjervis@ljervis.plus.). Not all simulium blackfly adults bite humans so we can hope that we have a non-biting species. The stomach contents of the grayling from the lower Alyn (Figure 2) suggest that blackfly may be present and eaten so artificials that provide effective imitations may work on the RGFFC waters, as might chironomid (buzzer) imitations. My own flies, for what they are worth, include three of the late John Goddard’s flies (ref. 2) – the Goddard Smut, the Black Gnat and the Suspender Midge Pupa, all in the smallest size I can tie – 18, along with a small Klinkhamer and the Griffith’s Gnat, also in size 18. Smaller sizes would probably be better but are beyond my tying ability. Any body colour will do, as long as it’s black, and for the Klink and Griffith’s I use a grizzle hackle. Earlier in the season, a Partridge and Orange Spider (16) works well for me.
Catch Returns
Another potential source of data comes from returns of catch data, ideally containing information on where the fish were caught, when and on what fly – even dry, emerger, wet or nymph/shrimp, would be fine. Unfortunately too few members submit catch returns to draw any sensible conclusions.
The fact that firm conclusions are difficult to draw from any one set of data confirms the need for triangulation sampling:
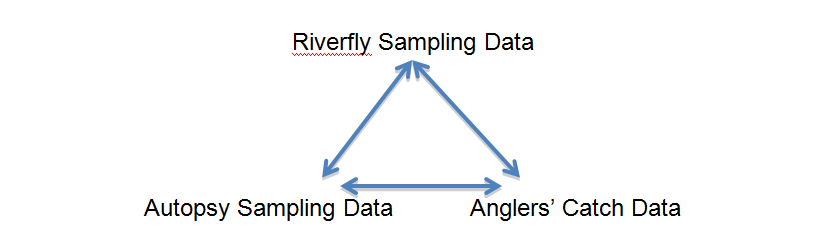
Only by doing this can conclusions from one data set be confirmed by reference to the other data sets – please send in your catch data, it is invaluable!
Waterweed
One of the essential features of rivers as far as providing both food and shelter for fly larvae is waterweed – and there is precious little in our stretch of the Alyn. Numerous attempts over several years have failed to establish ranunculus (water crowfoot), including the attempt to transplant into the mill leat this year. The water seems clean enough, in spite of sediment, waste water inflows etc and the establishment of blackfly is a pointer in this direction as both larvae and pupae require clean water. The high flows experienced by the Alyn and the fluid bed are most likely to blame but success at establishing ranunculus should pay dividends (see ref. 3).
Efforts to improve the RGFFC waters for both current and future members, to say nothing of the fish and other wild life, have depended on the time given by a small group of ageing work party volunteers. Everything from balsam bashing, removing fallen trees, stabilizing banks, cutting back bankside vegetation to planting ranunculus helps to establish better conditions for river life. If you have the interests of the club waters at heart and want to put something back into fishing for all the enjoyment it has provided over the years – don’t just grumble about the lack of fly life and the unwillingness of fish to rise to a well presented dry fly, join the working parties organised through the club web site. We might just get back to good rises of BWOs and other upwings with corresponding surface feeding activity by the fish!
A problem about the fly life on the Alyn? The simple answer is probably – a change in species? – yes; a change in total fly biomass? – almost certainly not, but we will need to review our artificials and change them to match the changed fly populations.
Acknowledgements
Thanks are extended to Professor Jaimie Dick and Helene Bovy of Queen’s University Belfast for comparative analysis of the predatory behaviour of Alyn gammarus. Thanks also to Eddie Wilkinson for the trout autopsy photograph and to Mark Pierce for letting me help with Riverfly sampling. Thanks also to the working parties for their untiring efforts on behalf of the RGFFC and the river Alyn. Many thanks to Mark Pierce and Howard Stevens for reading and commenting on the draft of this paper. The author takes full responsibility for any bullsxxt contained therein!
References:
- “The Alyn Anglers: A Century of Rossett and Gresford Flyfishers”, Paul Leng.
- “The Trout Fly Patterns of John Goddard”, (2004), The Lyons Press, Guilford, CT 06437.
- http://www.wildtrout.org/news/video-electro-fishing-survey
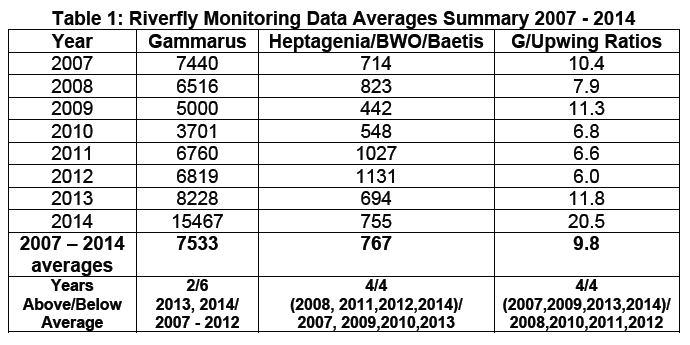

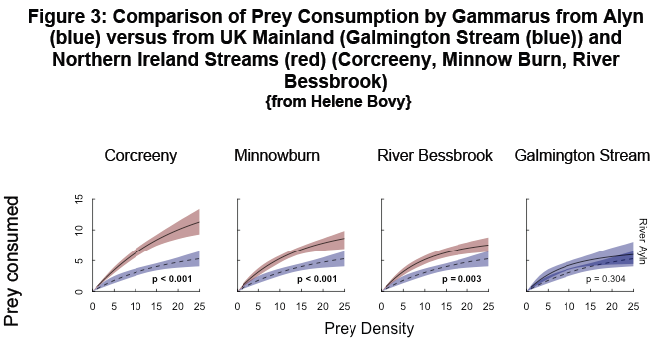

Les Jervis
Oct. 2014

A Day out with Geoff Clarke (Orvis shop Tarporley).
I have known Geoff many years through his job at Orvis and heard that he was leaving the area. A while ago he had a stroke that has stopped him driving, although he did return to work at the store in Tarporley.
We spoke on the phone and arranged a final day on the river, as he had only fished it once in his years in the club (and that was when he did a talk at our first “River Day”). I picked Geoff up at his home in Hoylake at 09:00 and as a 1st point we went to the Sewage field, as he had not fished for some time, so needed an area to get back into river fishing that was not too difficult. He started at the downstream end of the Kingfisher Pool and worked upriver.
.jpg)
.jpg)
A few fish were missed while he returned to form. As no fish were showing he had started with a single GRHE, but when he arrived at the Sewage Pool the surface activity proved that there were a few good fish on the top. A change to a parachute dry was needed. Although this pool can be very frustrating Geoff had a few “takes” to the fly before he finally got his timing right and a lovely Brown came to hand.
.jpg)
.jpg)
We then had a break for lunch at The Alyn Pub, where Geoff told me of his future plans. His is moving down south, but is remaining with Orvis and will be working part time in their local shop. He has sold his house here and will leave the area in early July.
.jpg)
.jpg)
Following our break we then made for September Cottage as Geoff felt more confident about wading our very pebbly river. Starting at the lower end by the wood he worked his way upstream, now casting both left and right handed, even the fish were impressed.
As there were no rises here he was using a mix of GRHE and PTN, both were managing to interest the fish, Geoff did however find that their speed of take, did not always result in a hook up – just too fast!
.jpg)
.jpg)
The pool just below the bridge was a prime example. I was watching from above and could see Grayling turning on the gravel bed and in the deeper run, but they were hitting the fly far too quickly for Geoff.
We went as far as the old packhorse bridge, Geoff fished both sides having fish take in both areas. By this time I had converted him to two flies and an indicator!!! It seemed to work well, as he managed both Browns and Grayling to the duo set up.
.jpg)
An excellent day was had with a few fish caught in difficult circumstances and very warm weather. All that can be left to say is Geoff, enjoy your new home and remember the River Alyn.
Eddie Wilkinson
June. 2014
(NB. Eddie forgot to set the time/date on his camera. He didn't take Geoff fishing in the early hours on the January 1st last year.

Intended Publication: “Grayling” Journal of The Grayling Society.
DRAFT 3: Jan.2014
Gammarus pulex – our very own killer shrimp
Les Jervis
From American mink, Himalayan balsam, American signal crayfish and Rainbow trout to Dikerogammarus villosus - the invasive Ponto-Caspian amphipod (shrimp) - we are all pretty familiar with the ‘alien invasion’ and its impact on the aquatic environment. Some of these ‘aliens’ are more welcome than others. The rainbow trout in particular has helped to revolutionise trout fishing in the UK. There are many more ‘invaders’ than these of course, but in most cases their impact on the aquatic environment is either unknown, minimal or nil. There are several potentially serious ‘alien invaders’ now in the UK with the bloody red shrimp (Hemimysis anomala), the amphipod Echinogammarus berilloni and water hyacinth (Eichhornia crassipes) being just a few.
Aliens or invited guests?
I’ve been putting the words alien and invader inside inverted commas because the majority have been quite deliberate introductions to the UK. Some have been here for so long that they are accepted as naturalised. Probably, the term ‘non-native introduction’ applies best to the majority of species. That they have been introduced deliberately makes them no less of a danger to the environment. They can eliminate native species, reducing biodiversity drastically. In addition to any immediate effects, introduced species often carry invisible hitch-hikers – fungal, bacterial or viral diseases or parasites that can wreak havoc with native species. The effects of crayfish plague, introduced on American signal crayfish, on our native white-clawed crayfish being a good example. The larger non-natives are easy to spot but we have no idea how many micro non-natives there might be.
Rapid spread
The rate of spread of successful non-native introductions can be frightening, as anyone who has spent time helping to clear Himalayan balsam knows. Balsam is an attractive plant that walkers, bees and some bee keepers love but it wipes out normal riverside plants and it dies back in winter, leaving river banks wide open to increased erosion from winter storms. Dikergammarus villosus, the killer shrimp, is increasing in UK waters where it can rapidly eliminate the native Gammarus pulex and other macro-invertebrates, including fly larvae. Not that the fish mind, is a large and juicy mouthful for trout and grayling alike! The trouble is that, if the spread in the UK is as rapid as it has been in mainland Europe, the native Gammarus pulex and other aquatic invertebrates have a bleak future.
Dikerogammarus in Europe
Much European work on Dikerogammarus spread has been done in the laboratory of Gerhard Maier at the University of Ulm in Germany. The main spread of Dikerogammarus villosus in Europe came with the opening of the Main – Danube canal in 1992. The amphipod had been found in the German Danube in 1992. It invaded the Main in 1993/94 and the Rhine in 1995. In 2002 it was recorded for the first time in the north-western part of upper Lake Constance. Within two years it had colonised the entire upper lake and had spread by 2004 to the lower Lake Constance. It is, by far, the most successful invader of the waters of Southern Germany. Its prolific breeding is a major reason for its success, with each female producing clutches of 100+ eggs during the spring and summer. This compares with probably fewer than 30 eggs per clutch from G. pulex females. Where the latter have some advantage is that they continue to reproduce during the colder months and are able to regain some lost ground. However, in addition to its successful breeding strategy, D. villosus has strong predatory behaviour. Although classed primarily as a detritivor, feeding on dead plant or animal material, its success is due to its being a flexible omnivore. Like man, rats and cockroaches it can, and will eat, almost anything including other aquatic macro-invertebrates. It is even cannibalistic when the opportunity or need arises. Not only does it eat G. pulex, it drives them out of house and home as well. By displacing the smaller G. pulex from its refuges under gravel and stones it exposes them to other predators such as fish so the native G. pulex has literally nowhere to hide. This appears to be particularly true where rivers and streams lack aqueous vegetation, so swans and other vegetation-consuming water birds add to the pressure on G. pulex, further exposing them to predators. The same is true for other aquatic macro-invertebrates that use plant life as refuges and feeding sites.
A re-assessment of Gammarus pulex
All this might sound like G. pulex is a totally innocent potential prey item. Nothing could be further from the truth. Since the early 1900’s gammarids have been known to feed on carrion and have been raised in laboratories on items such as chopped earthworms. In 1924 Gammarus duebeni was identified as having cannibal tendencies. That observation is particularly interesting since G. duebeni is the native gammarid in the Isle of Man and Northern Ireland. By 1954 G. pulex, introduced into the Isle of Man from mainland UK waters was shown to eliminate G. duebeni rather than form mixed populations. Between 1960 and 1995 G. pulex was shown to consume caddis larvae, chironomids, tubifex worms, Ascellus aquaticus, rainbow trout eggs and mosquito larvae. It was also shown to be cannibalistic again with males have a preference for moulted females. In 1997, Jamie Dick and his colleagues from Queen’s University Belfast proposed a re-evaluation of G. pulex after showing that, when it was introduced into Northern Ireland, it eliminated the native G. duebeni from the river Lagan. By 2002, G. pulex was known to predate on Baetis rhodani, adding to its known prey range and giving cause for concern for those of us whose waters have poor hatches of upwing flies but large populations of G. pulex.
In addition to its unwanted predation on upwing fly larvae, G. pulex has helped to fight off a potential invasion by the American amphipod Crangonyx pseudogracilis. Even the less predatory G. duebeni can out-compete the C. pseudogracilis. Professor Jamie Dick, who studied the system, aptly titled his paper “Predators vs Alien”. Nonetheless, G. pulex can lead to population destabilization of native prey items.
Does Gammarus pulex contribute to the decline in fly life?
Many rivers in the UK have suffered from major declines in aquatic fly life. Much of the decline is probably due to increased environmental degradation from water abstraction through to diffuse pollution but G. pulex could be a factor in some cases.
G. pulex and other gammarids are most common in chalk streams or where the water has a relatively high pH. My local river, the Alyn, is a tributary of the Welsh Dee but, unlike the Dee, the Alyn springs from limestone so is more alkaline. In theory, the river should have good populations of upwing fly larvae but as on many other rivers, these seem to be in decline. One of our club members, Mark Pierce, has seen G. pulex attacking and eating Baetis rhodani larvae while he has been doing invertebrate sampling so, locally, we have evidence of predation. The extent to which predation affects overall fly life and whether this is a major issue on other rivers, remains unanswered.
Water quality
The Alyn has upwards of a dozen waste-water treatment plants discharging into it, the furthest downstream being near Gresford on the Rossett and Gresford Fly Fishing Club (RGFFC) waters. Invertebrate monitoring for the Riverfly Partnership gives a relatively healthy Angler’s Score Index (ASI) of between 10 and 12 for biodiversity, well above the trigger level of 4 for severe water quality problems.
The fish appear to be very healthy. We don’t have much data from autopsies on fish diet in the Alyn but we know that in the sewage discharge pool, fish can be full of chironomid larvae (see “Grayling” Autumn 2011, page 36). This is an interesting observation as chironomid larvae are an easily-caught favourite prey item for gammarids. The number of sewage discharges and the numbers of flying midges, points to very high chironomid populations throughout the river. One consequence of a large chironomid population is that it could help boost the G. pulex population. Unfortunately, the Riverfly Partnership sampling does not include estimates of their population numbers.
Usually, chironomids are associated with slow-flowing or still water and they can thrive at low oxygen levels. An initial thought was that the discharge pools might have low oxygen but the numbers of avidly feeding fish seemed to argue against this. Kick samples at the tail of the Gresford works discharge pool showed just as many G. pulex as above the pool so the river does not seem to have discharge effluent with high biological oxygen demand (BOD).
A major concern, particularly with the number of sewage treatment plants, is that chemicals in the discharges might affect the behaviour of the G. pulex. Certainly water containing low levels of synthetic hormones can cause increases in the female:male ratio and stimulate breeding, causing dramatic increases in the numbers of neonate and juvenile gammarids. So, far from reducing gammarid numbers due to a lowering of water oxygen levels, the sewage discharges may be doing the opposite.
Whilst sewage discharges are more rigorously controlled than in the past, the nature of sewage has changed. There are vastly more pharmaceutical and ‘personal care products’ in sewage than there were. Many of these are not fully degraded before discharge into rivers and streams. These micro-pollutants can have significant effects due to chronic exposure and the cocktail of compounds is complex and ever changing. Monitoring or predicting biological effects is almost impossible. The chemicals are usually colourless so visual monitoring of outflows does not give any immediate cause for concern. Levels are usually low so there is rarely any immediate toxic effect and BOD is low. However, it is known that Dikerogammarus villosus can tolerate lower oxygen than G. pulex so if the Alyn does get invaded by D. villosus, the outlook for the native G. pulex is poor.
The number of sewage treatment plants is unlikely to diminish. The volume of sewage treated will only increase as the human population increases. Continued improvement in the quality of water discharged must be hoped, and campaigned for. Continuing to simply discharge treated waste-water into rivers that are probably already suffering reduced flow as a result of abstraction, is not a sustainable option. Discharge volumes will continue to increase, as will abstraction. Increasing heavy rainfall will cause further problems as storm flows discharge directly into rivers. Treated outflows need longer to trickle through an area designed to allow complete biodegradation and filtration before final discharge into the river. Some form of reed bed or sustainable drainage system (SuDs) would, I feel sure, help considerably but I don’t know how feasible building and maintaining such a system between the outflow of every waste water treatment plant and its receiving river would be.
What can we do?
Although we have a biodiversity problem on the river Alyn (as elsewhere), the fish are healthy and the fishing is good. It might be tempting to do nothing and hope the situation sorts itself out. There is always a risk in trying to manage an ecosystem and there is a real possibility of making things worse. My personal belief (you can tell I’m an ex-academic) is that more research is needed. On the Alyn we need to know:
- the underlying reason(s) for the high G. pulex population numbers,
- the full extent of predation on fly larvae,
- the pattern of fly life decline over time,
- the main fish diet items (from autopsies through the season)
- the extent to which fish add to the problem by eating those fly larvae driven out of their refuges by predation from gammarids,
- the contribution made by chironomids to G. pulex prey items,
- the role played by the numerous sewage discharges,
As far as the river ecology is concerned, we should be able to come up with a fisheries management plan that tries to deal with the situation and slowly improve it. Rather than reducing the gammarid population quickly, a slow reduction whilst trying to increase upwing fly numbers might maintain insect biomass and, therefore, food for the fish.
Trying to increase fly life by transplanting eggs against a background of heavy predation by both fish and gammarids may be premature but it is worth putting in some well-placed fly boards in addition to the felled trees etc already placed to provide refuges for small fish and invertebrates. These would increase the locations where our remaining Baetis can lay their eggs. If the boards were placed so that they were off the riverbed, away from Gammarus predation, our BWOs might stand a chance of making a comeback. Baetis are, after all, probably more pollution tolerant than most upwing fly larvae so they will be a good species to start with.
If we keep bashing the balsam, more natural bank vegetation should re-establish and provide daytime refuges for those BWOs etc that survive to hatch. We might even get a virtuous circle of regeneration going.
Actions by an individual club is always going to be less than ideal. What happens upstream of club waters in particular is always going to have a significant effect. Even what happens downstream will affect upwards migration, both of migratory species and grayling, but also aquatic flies, many of which migrate upstream for mating and egg-laying. Much better a whole river catchment-based approach such as is now proposed (by e.g. DEFRA etc) and has been trialled on the Eden, the Wensum and the Hampshire Avon and which seems to be in prospect for the Alyn.
However, better some immediate, but carefully considered, remedial action and more research to try to fully understand the system than wait for an Alien Invader to come along and clear out the G. pulex for us! The fish probably won’t care, bigger shrimps will be a bigger mouthful, but the diminished biodiversity and the accompanying loss of aquatic fly life will make being by the river a poorer experience.
Acknowledgements
I would like to thank Howard Stevens and Mark Pierce of the RGFFC for stimulating my interest in Gammarus pulex and for reading the draft manuscript, Professor Jamie Dick of Queen’s Unversity, Belfast for sending me reprints of some of his papers, and Philip White, ex-river keeper of the Haddon Fishery on the river Wye in Derbyshire, for very helpful discussions and suggestions.

Dikerogammarus villosus
Ponto-Caspian alien invader.
More predatory than, and displacing, G. pulex

Gammarus pulex
UK mainland native species but an alien introduction to the Isle of Man and Northern Ireland. More predatory than, and displacing, G. duebeni

Gammarus duebeni
Native species in the Isle of Man and Northern Ireland. Present in brackish waters in mainland UK

Crangonyx pseudogracilis
American native. Present in Europe but out-competed by both G. pulex and G. duebeni

Partly devoured upwing fly larva. Note bits of legs, tail and antennae missing. Photo from Mark Pierce taken 10 minutes after collecting mixed G. pulex and fly larvae sample.
Main References
MacNeil C, Dick JTA, and Elwood RW, (1997),Biol. Rev. 72, 349 – 364,
The Trophic Ecology Of Freshwater Gammarus Spp. (Crustacea:Amphipoda):
Problems And Perspectives Concerning The Functional Feeding Group Concept.
Kelly DW, Dick JTA and Montgomery WI,(2002), Freshwater Biology (2002) 47, 1257–1268
Predation on mayfly nymph, Baetis rhodani, by native and introduced Gammarus: direct effects and the facilitation of predation by salmonids
Kelly DW, Dick JTA, and Montgomery WI,(2002),
Hydrobiologia, 485,(1-3) 199-203,
The functional role of Gammarus (Crustacea, Amphipoda): shredders, predators, or both?
MacNeil C, Dick JTA, Johnson MP, Hatcher MJ, Dunn AM, (2004)
Limnol.Oceanogr 49(5), 1848–1856
A species invasion mediated through habitat structure, intraguild predation, and parasitism.
Krisp H, Maier G, (2005),J.Limnol64(1): 55-59,
Consumption of macroinvertebrates by invasive and native gammarids: a comparison.
Watts MM, Pascoe D, Carroll K (2002)
Environmental Toxicology and Chemistry, 21(2), 445 - 450
Population responses of the freshwater amphipod Gammarus pulex (L.) to an environmental estrogen, 17α-ethinylestradiol.
Kinzler W, Kley A, Mayer G, Waloszek D, Maier G, (2009)
Aquatic Ecology, 43, 457 – 464,
Mutual predation between and cannibalism within several freshwater gammarids:
Dikerogammarus villosus versus one native and three invasives
Dick JTA, Armstrong M, Clarke HC, Farnsworth KD, Hatcher MJ, Ennis M, Kelly A, and Dunn AM, (2010) Biology Letters 2010 6,
Parasitism may enhance rather than reduce the predatory impact of an invader.
Dick JTA, Alexander ME, MacNeil C, (2012),
Biological Invasions DOI 10.1007/s10530-012-0287-9
Natural born killers: an invasive amphipod is predatory throughout its life-history.
MacNeil C, Dick JTA, Alexander ME, Dodd JA, Ricciardi A,(2013)
NeoBiota19: 1–19
Predators vs. alien: differential biotic resistance to an invasive species by two resident predators.
SuDs:
http://www.sepa.org.uk/water/water_regulation/regimes/pollution_control/suds/suds_explained.aspx
Demonstration Test Catchments- building our capacity for catchment management:
http://www.euwfd.com/SH100924c_Demonstration_Test_Catchments.pdf
http://www.lwec.org.uk/activities/demonstration-test-catchments
National Demonstration Test Catchments Network:
http://www.demonstratingcatchmentmanagement.net/
Les Jervis
Jan. 2014

Living with Indicators
Now most fly fishers will at least be aware of this method of fishing, agreeing with it may be a different matter. On a personal note, for stillwaters I have very little use for them, but if it is within fishery rules and conditions are right, I have no problem using "some" types. For river fishing, especially upstream nymphing the difference it can make is astounding.
My local water is the River Alyn near Wrexham, North Wales. It is a small rain fed tributary of the Welsh Dee. Not a deep water, with very few places over 3' deep - for most of the clubs length (3 - 4 miles) only as wide as a single track road.

There are a lot of folk who class the use of indicators as "float fishing" or de-skilling the art of fly fishing. If you have perfect eye sight and can watch your line in shade and bright sunshine to see, almost microscopic movements, then read no further. For those less able, it does give some aid, for example:-
A few seasons ago a new(ish) member approached me on the banks, to ask how I was going on, I replied that I had caught several fish to the upstream nymph. Admitting that he was not catching he asked if I could show him how:-
I was fishing a single size 14 weighted PTN spider with a small butterfly indicator (the sticky fold over ones) set at about 3' (water depth about 2'), this initially horrified him. I looked at his set up, it was not too dissimilar from mine, with a single size 12 weighted PTN. I asked where he had fished and one pool was not far away, so we went back to it and I used my set up to fish the same pool. Even though I had a small indicator on, the takes were still hard (in some cases) to see, just a stop, for the briefest moment, these I tried to show, before attempting a hook up. The first two fish I did catch, he did not see the takes! I passed my rod to him and he tried. I pointed out when he had had takes but his reactions were a bit slow to start with. He eventually connected to about the 5th and 7th fish! As he commented after, on his set up he would not have seen any of the takes. This was on a pool he had already fished though and we still had 4 fish out of it and at least half a dozen missed "takes" between us!
By setting the maximum depth it is possible to put the nymph in the "food" zone, but only by knowing your river.
While dry fly is my preferred method of fishing, on a rain fed river this is not always possible. Across and down on a mostly shallow river, that is less than 5m wide (less in some places) is not effective, so by elimination it has to be the upstream nymph. Most of the time a single fly tied to a 5' section of tippet is all that is needed, with the indicator mounted at the appropriate depth. But to cover high, fast water something different is needed, below are the details.
Early season and when the fish refuse to rise are ideal times to try this method. When the river is running up and fast a second weighted fly is sometimes needed to get to that all important "feeding" zone. Sometimes it can be that the fish will only take small flies just above the riverbed. At this point it can be a heavyweight fly on the dropper (leaded B175 size 10ish) with a small (B175 size 16 or Fulling Mill Nugget size18) on the point, these flies can be less than 12" apart. The theory being that the heavy hook gets the smaller fly down into the feeding zone and the lack of weight on the point fly gives it a more natural movement in the river. Problems arise when too heavy a dropper is used, the indicator sinks or the bottom keeps snagging. This can mean several changes of fly to suit the river levels as you move runs.
Typical set up for 2 flies - a weighted size 10 pink shrimp and a beaded size 14 PTN (ish)

Pre built ready to store - connecting line goes around from shrimp (hook bend) to nymph eye (course fish rig holder).

Storage for a twin fly set up

5 rig set up with two twin flies in each gives 10 sets. Simple to tie into a 3' to 5' tippet section from the fly line. Saves time and effort on the bank (or even in the river). It can be the same set up 10 times or various options of colour, weight or even dry and wet flies.
A Typical River Set Up
The foam indicator is positioned along the 3' to 5' section at a position to suit the depth to be fished (all my local river levels fit within this range)
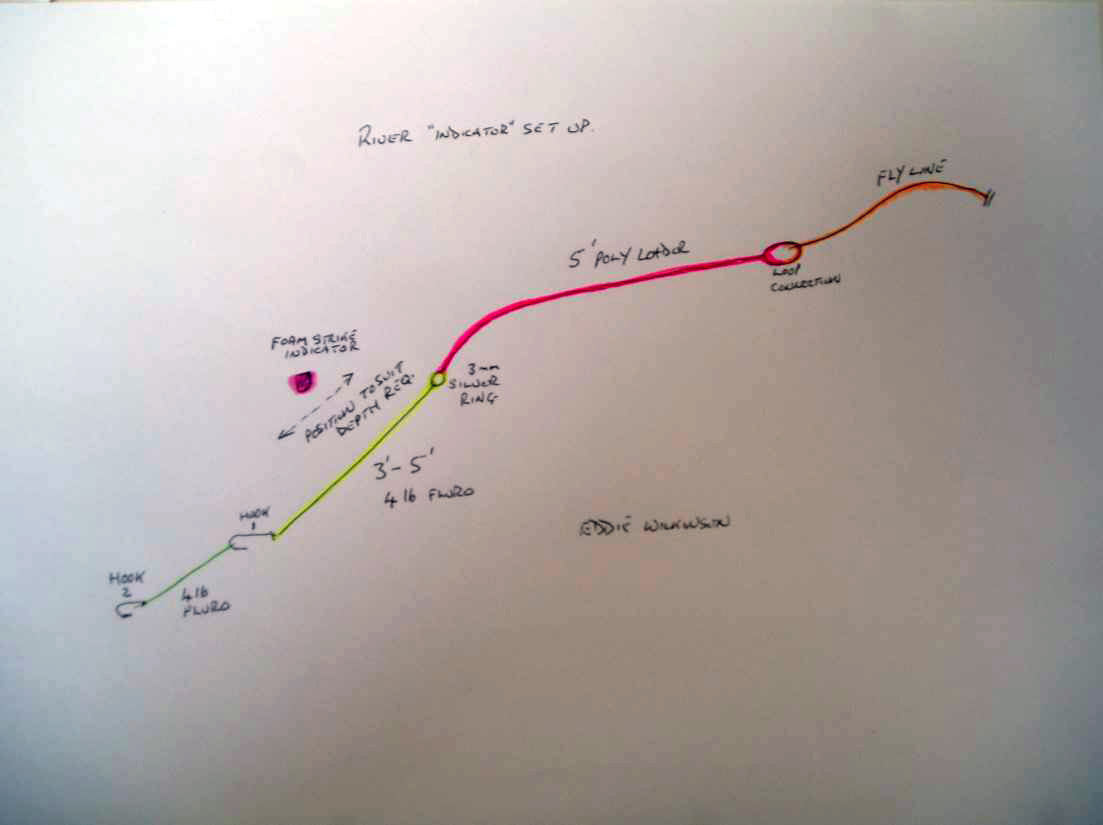
While not to every ones "cup of tea" where allowed it certainly helps. It is just another method along with Czech nymphing, French leaders, leader to hand or even Tenkara. If you haven't tried it yet, be brave, have a go, you may even find it helps.
Eddie J Wilkinson
March 2013
Go back to the top

Mustela Vison:-

The mink, also known as the feral mink, is a chocolate brown stoat like animal, often mistaken for otters because of it's aquatic life, but are much smaller. Well established in England and Wales now, and a fair few in Scotland, usually found on rivers, lakes and any fairly large water course. Although taken by buzzard or fox when they can catch them, their main predator will surprise most of you, it is the otter. They not only discourage the mink, they kill and eat them. Mink carcasses been found in and near otter holts but a study of their droppings or spraints have revealed so much evidence, that it is now deemed to be its main predator. But the mink on our river have another predator far worse than any otter. Over the years I have trapped most everything you can eat in this country and most pest species, but most importantly, members of the mustelidae family - the weasel family to you and me. This includes the mink. I have used most types of trap, mainly wire traps, drop traps, cage traps and spring traps. Spring traps are the favourites of most hunters and game keepers. These should only be used by the experienced, for they are indiscriminate unless used by the right people. I was taught from an early age the importance of using the right trap for the targeted species, for no hunter, or man for that matter, worthy of being called a man, kills indiscriminately. Ever since I watched my uncle and my dad set their first trap as a young boy, I was never allowed to touch them until I could recognise the different characteristics of every animal and bird we were likely to target, how and where they lived and fed, and also to read the land like a map, determining where to set the traps. I have described in a previous article, how to read a river in the same way, well, the same rules apply to the land in this context. Knowing your prey has never been more important.
Most animals are territorial but they frequently cross each other's territory, so it is important to know the difference between the tracks. Being able to read the land, I have an idea whose territory it is and being able to differentiate between the tracks, I know who the visitors are.
Which brings me to the river and its population of mink. People walk the river and see tracks on a sandy beach or muddy patch next to the water and assume they are the tracks of a mink - this may not be the case in lots of instances. The topography or natural characteristics of a river bank, make it an ideal habitat for a whole myriad of species, especially prey species, like rabbit, bank vole, shrews, rat and field mouse and others. These in turn are predated on by weasels, polecats, stoats and mink, to name but a few. Now, are you sure those tracks you saw are mink?
Let's look at those tracks in more detail. All of the mustelids have five toes but the most obvious thing to look for is the overall size of the track. You wouldn't mistake an otter for a weasel, simply because of the size. The tracks most commonly mistaken for mink are that of the stoat, they are very similar in size but like all tracks, there are things to look for. Distinct claws give the toes a pointed appearance, often with webbing between pads, but not always. Now the heel pads especially on the back feet have four lobes. But on the mink, the two central lobes are larger. It is this larger heel pad that distinguishes mink tracks from those of the stoat, despite the similar overall size. The other method I use for locating mink is to look for droppings, of course, knowing where to look and how to recognise them helps.
As with most animals, the deposit of their droppings are used for communication and to establish territories, so they are strategically placed. The otter and the mink wouldn't want it to get washed away, so they pick an elevated vantage point, like a log or a big rock. Mink droppings resemble that of the otter but there is one difference you will never forget once experienced.
Otter spraint has a sweet, lavender like smell to it but mink droppings are the complete opposite. It emits a very foul odour. If in doubt have a smell, as I have said, you will never forget the smell of mink. In fact, I remember doing just this one day and was so engrossed in what I was doing, that I failed to notice a figure standing on the riverbank looking down at me in disbelief. It must have seemed to him that I was kissing the rock but with a strange look of amusement in his eye, he listened intently as I explained what I was up to. I don't think he was completely convinced as he gave a chuckle and carried on his walk down river.
Now, there has already been a brief mention of some of the traps that I use. On my shoot, I use spring traps, set in tunnels but I restrict the tunnel entrance to ensure that I only catch mustelids and rats, for they will all kill my partridge and pheasant poults. For nuisance birds like magpies, some corvids and brown owls, I will use swing door Larson traps. and for squirrels I will use cage traps. In addition, I always have my trusty gun.
I would never eradicate any of the species mentioned to the point of extinction, for an ecology could not survive without a healthy balance of both predator and prey.
On the river, to catch mink, I use live catch cage traps for this very reason. If I catch a non target species, I can just let them go unharmed. A well set spring trap solves the problem of dispatching the animal as it kills instantly. With a live catch trap, most people will shoot the animal. My own method is even simpler. I put on a chain mail glove under a welding gauntlet, reach in, grab the animal and use a priest. Mad I know but simple and I've never been bitten yet. On the subject of where best to place the traps, all I will do is to refer you to my comments on my knowledge and experience of reading the land and knowing the habits of my prey. That's all I can say on this matter. I have had a couple of traps go walkabout and as they cost me fifty quid each I must be as secretive as the mink regarding their location.
My reason to write on this subject was prompted by comments on the forum by a new member who seemed to think that the mink are allowed to roam free on our fishery and that there was no one who knew how to catch them. I hope the above reassures him and any others who were not aware that I've been doing the business for many years.
Peter Hayes
Can you now identify the tracks?


Go back to the top

Creeping through the undergrowth............
Here am I, creeping through the undergrowth, listening to a bird, hearing a calf call its mother as it rejoins the herd, discovering an orchid, watching a busy insect or sometimes just sitting on the river bank, observing all around me and reflecting on nature. But what am I?
I am a fisherman. Everyone has their reasons to go fishing but for me, it's understanding the whole environment that makes a person a better fisherman than somebody that just goes to catch fish. Let me explain as I am not just a fisherman but a hunter and the first rule of any type of hunting is to know your prey; their habits, habitat and their defences.
Every species of animal has a number of defence mechanisms, whether it is sight, smell, hearing, vibration detection or just plain flight. Once all these are understood, the prey can be approached almost undetected. If I want to approach a rabbit or a deer, the first thing I do is to keep out of sight. I also make sure I am down wind - keep the wind in my face. This has two functions, it blows smell away from the prey and any sound made is carried away by the wind.
But what has this to do with fishing? Well, the trout has three very acute senses, sight, smell and sound in the form of vibration through the water, which is picked up by the trout by sensory organs along its lateral line. Look for a rising fish, then tap two stones together under the water and see how long it continues to rise.
Remember what was said about the importance of the wind, in the case of fish, the current of the water is our wind, but we must also look at the position of the sun. We must not cast a shadow over the fish as it will see us. The current, if we approach from downstream will carry any smell or vibration away from our prey. In this way it is possible to get closer and into a better position and yet still remain undetected. Trout have good eye sight and because they lie facing upstream where the majority of its food comes from, any shadow or movements would be detected and send it darting for cover.
This is only part of the rules. Know your prey, the next, is to find out as much as you can about its habits, its diet, what time it leaves its lair and what's the favourite feeding position. We will cover this later but now, know your environment.
What is a river? A silly question? We have all walked a river; we know what kinds of fish it holds. Why? Because we have probably been told. However, if you come across a stretch of river and have no knowledge of the water, would you know what fish are likely to be there, which, of course determines what method to use to catch them. It is therefore prudent to provide a biological description of a river.
The moving waters of streams present quite different living conditions from those found in still waters and ponds. What is more, the conditions vary as one moves up and down the stream. It is possible to divide a stream into several regions according to its animal and plant life.
The upper reaches of a stream form the headwaters. These are usually on high ground and are normally shallow and fast flowing. There is plenty of oxygen here, and the main problem for the resident population is to avoid being swept away. The only plants that manage to grow here are algae that attach themselves to the rocks. A few small snails crawl along the bed of the stream but the main animals are the young stages of various insects; flattened nymphs of some mayflies and stoneflies cling to the stones and nibble away at the algae. There are also caddis larvae, which make themselves little cases of sand or plant grains.
The headstream gradually merges into the 'troutbeck' region. The slope is not as steep here but the bed is still rocky and the water runs quickly. A few patches of water crowsfoot may grow on the stream bed, but there is little other vegetation apart from the algae. The flattened insect nymphs of the headstream also live in this region, together with various snails and the river limpet. The limpet has a conical shell, but is not closely related to the limpets of the sea shore. Fish make their appearance in the 'troutbeck' region. They are either strong swimmers such as the trout or they hide among the stones and boulders - the loach and bullhead are among these bottom dwelling species. All feed on insect nymphs or on adult insects that fall onto the water surface.
The next stretch of river is usually called the minnow reach. The current here is slower and a certain amount of sand and gravel covers the bed of the stream. There are plenty of water plants and animal life is also more abundant. There are many different kinds of water snails, dragonfly nymphs, mayflies, and other insects. The minnow is the typical fish, but there are also sticklebacks, lampreys, dace, grayling, trout and salmon. Another common insect is the fresh water shrimp, which darts over the sand and among the plant stems.
Below the minnow reach, the river gets deeper and slower. This is called the 'cyprinoid' reach, characterised by coarse fish such as roach, rudd, perch, pike, chub, bream and carp. The slower moving water deposits plenty of mud and plants are able to grow along the margins; worms, water snails and mussels are common in the mud. There is a certain amount of floating plankton in this stretch of river, especially in the weedy parts, but it is never as common as it is in the still waters of a pond.
There, that is a river. Now that piece of running water you came across becomes more recognisable as to what stretch of river it is, what is likely to be living in it, and so, provides a clue how to fish it.
This is the importance of the second rule of hunting/fishing; know your environment.
You have found your prey, the conditions are right, so you now have to catch it. Apart from shooting and the use of dogs, traps are used and whatever type of trap you use, the prey has to be enticed into it. This is the second part of the first rule, know your prey. For a trap to be successful you have to bait it and the best bait is something the prey wants! In the case of fishing, the hook is the trap and the fly is the bait. This is where it is essential to know the habits of your prey, its diet and when it feeds.
Trout essentially feeds on water borne insects, but it also has its predatory side, taking small fry of its own species, minnows, and elvers. However, insects are the main part of its diet, so this is what we need to take advantage of, giving them what they want. This method of trapping your prey is commonly known as fly fishing.
The two main types of fly fishing use surface flies or sunken sub surface insects. You will understand this a little later when I talk about entomology. But now we are looking at habits, diet and feeding times.
Firstly, where to fish. Well, in the case of a rising fish, the problem is solved. You can see where he is. The trout feeding on surface flies sips them as they float towards him. This is the rise. So you give him what he wants by casting a dry fly a couple of yards above where you saw the rise and letting it float over him, hoping it's to his liking.
In the case of sub surface flies, you are not casting to a rising fish as you don't know exactly where he is. But there are things you can do or not do! The first is not to cast aimlessly or blindly into a pool. Remember that I said that a trout is always looking upstream for the river to bring food to him. Look at or into the water and you will have plenty of indications to the trout's position. Other insects come floating by, or bits of debris. Where did they come from, which current brought them into the pool, which line did they take as they passed through the pool? Once this is established, you will know the likelihood of this being the food trail, and you can bet the trout know this too and this is where they are likely to be. So now you are not fishing blindly but with an education, which brings confidence in your casting and a definite advantage over someone who just chucks and chances it.
What have we established so far in this article? Well, you know your prey, you know your habitat, you know where to find your prey and where they are likely to be feeding, and more importantly you know how to approach your prey using stealth. Lastly you know their general diet. I say general diet, but you really have to know their diet in particular. The trout is a very particular feeder. When he is taking a particular fly, he will touch no other. On the other hand, he could be feeding on two or three different types of fly at the same time. This can only be established by observation and a good fisherman spends just as much, if not more, time observing than actually fishing. What are we looking for? We are watching for flies either floating down river, or taking off the river, the general colour and size of the flies and most importantly, are the trout taking them. Flies taking off from the water usually mean a hatch is on and this is the time for the fisherman to get out his fly rod. But what brings a hatch on? This is the time to examine that word again, entomology. This is the study of insects, in this case, water borne insects. The flies you see floating down the water with their wings held upright where actually born or hatched on the stream bed. They live as larvae or nymphs for as long as three years, or short enough to allow three generations in one year depending on the species. Most are vegetarians, one or two carnivorous. They live in the gravel beds, cling to the stones or bury themselves in the mud. Some will get washed away by the current and this is the time when fishermen will use a sub surface artificial nymph to tempt the trout. The nymphs go through various moults and will develop wing cases in which their developing wings are protected. Just before the final moult of larval skin, they come to the upper layers of water ready to emerge. This is when we use a wet fly sub surface imitations but with their wings tied back, for they are still enclosed in the larval sac. When they finally emerge on to the surface and this sac is discarded, they will float down stream holding their wings high to dry ready for their first flight, which is usually a weak effort. This is the time when we see the fish rising and therefore fish the dry fly.
So, there you have it. Are you a fisherman or a hunter? They are one and the same, following the same rules. Know your prey, their habitat, learn stealth and most importantly, practise observation and, as with all pursuits, be patient.
Whether for the experienced or inexperienced, there are always tips to be learned. However, some rules are set in stone from time immemorial. Learn them and they will stand you in good stead.
So, get out there and good hunting.
Peter Hayes.
Go back to the top
